Chapter 18: Q53P (page 915)
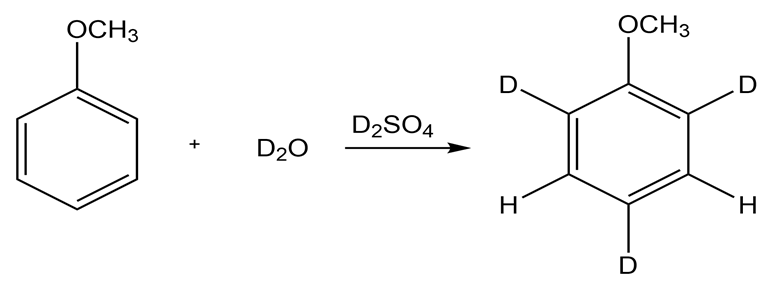
If anisole is allowed to sit in that contains a small amount of, what products are formed?
Short Answer
The product formed when anisole is placed in is,
Learning Materials
Features
Discover
Chapter 18: Q53P (page 915)
If anisole is allowed to sit in that contains a small amount of, what products are formed?
The product formed when anisole is placed in is,

All the tools & learning materials you need for study success - in one app.
Get started for free
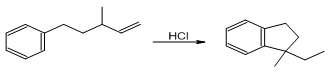
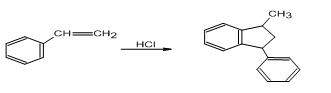
Propose a mechanism for each of the following reactions:
a.
b.
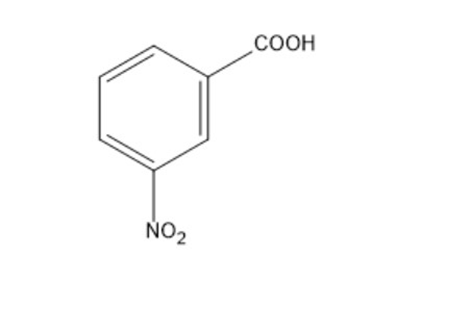
Show how the following compounds can be synthesized from benzene:
a)
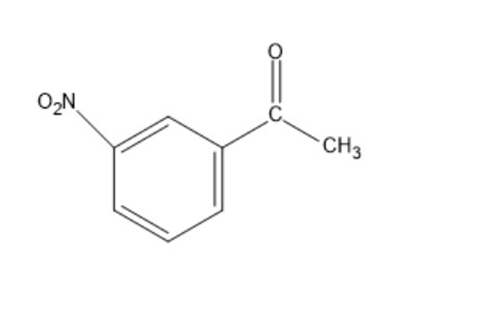
b)
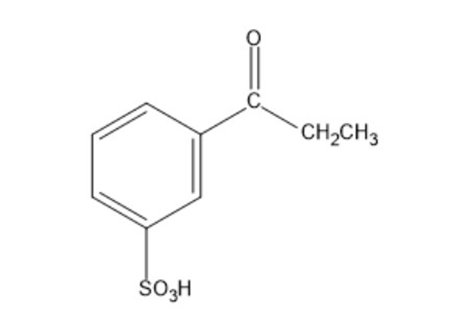
c)
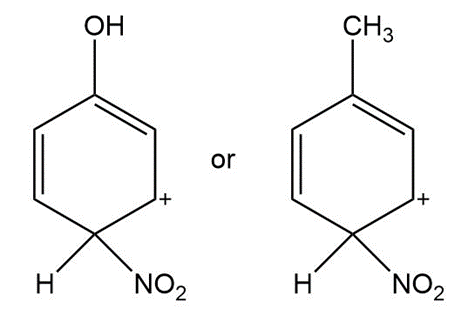
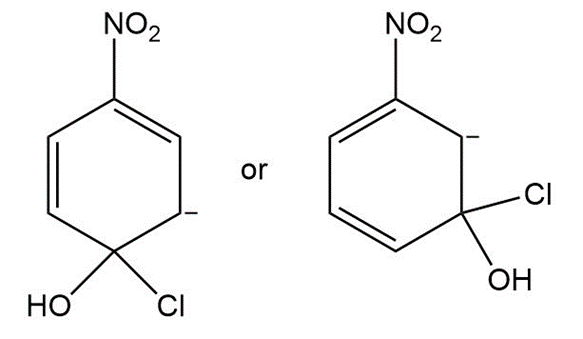
Question: Which is a more stable intermediate in each pair?
a.
b.
Question: Draw a structure for each of the following: a. 2-phenylhexane
b. benzyl alcohol
c. 3-benzylpentane
d. bromomethylbenzene
Question: The pKa values of a few ortho-, meta-, and para-substituted benzoic acids are shown below:
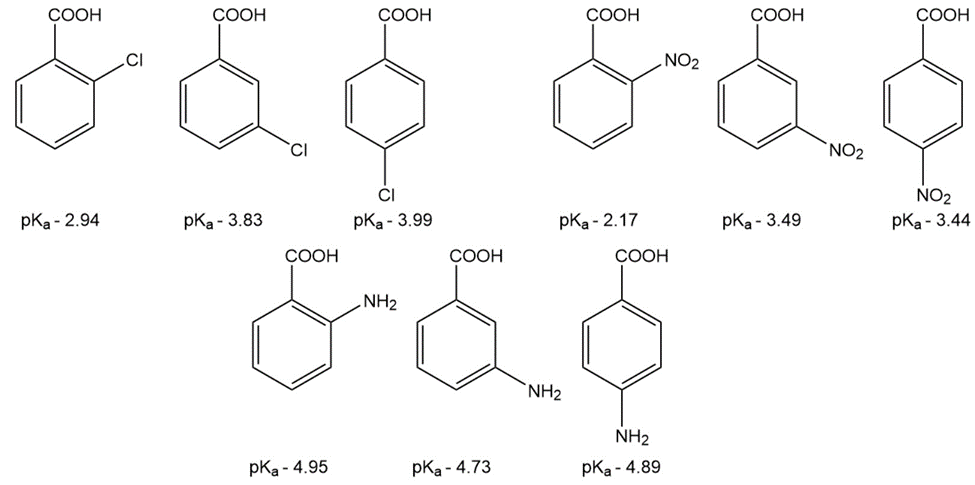
The relative pKa values depend on the substituent. For chloro-substituted benzoic acids, the ortho isomer is the most acidic and the para isomer is the least
acidic; for nitro-substituted benzoic acids, the ortho isomer is the most acidic and the meta isomer is the least acidic; and for amino-substituted benzoic acids, the meta isomer is the most acidic and the ortho isomer is the least acidic. Explain these relative acidities.
a. Cl: ortho > meta > para
b. NO2: ortho > para > meta
c. NH2: meta > para > ortho
What do you think about this solution?
We value your feedback to improve our textbook solutions.