Chapter 18: Q69P (page 917)
Does m-xylene or p-xylene react more rapidly with Cl2 + FeCl3? Explain your answer.
Short Answer
m-xylene will react more rapidly.
Learning Materials
Features
Discover
Chapter 18: Q69P (page 917)
Does m-xylene or p-xylene react more rapidly with Cl2 + FeCl3? Explain your answer.
m-xylene will react more rapidly.
All the tools & learning materials you need for study success - in one app.
Get started for free
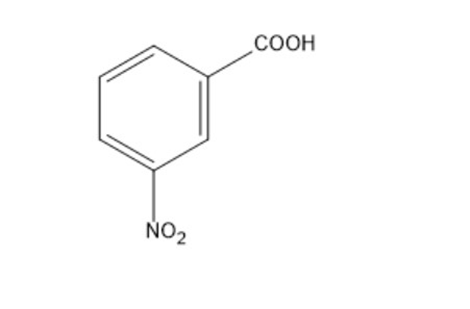
Draw resonance contributors for the carbanion that would be formed if meta-chloronitrobenzene were to react with hydroxide ion. Why doesn’t the reaction occur?
Draw the product of each of the following reactions:
a.
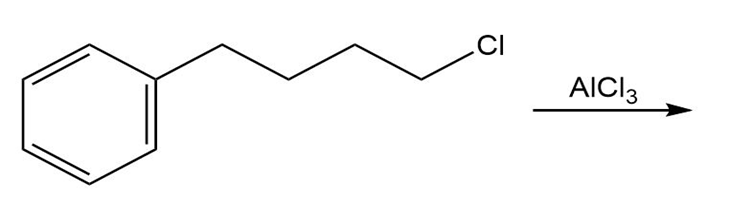
b.
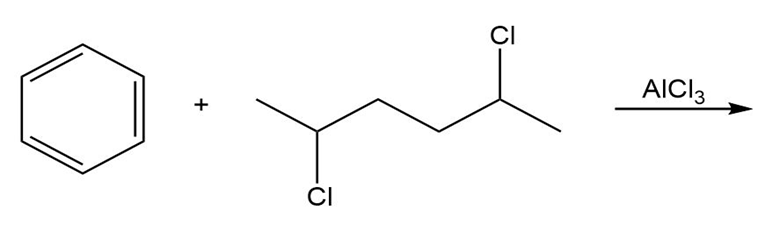
a. Does a coupling reaction have to be used to synthesize p-dipropylbenzene?
b. Can a coupling reaction be used to synthesize p-dipropylbenzene?
Show how the following compounds can be synthesized from benzene:
a)
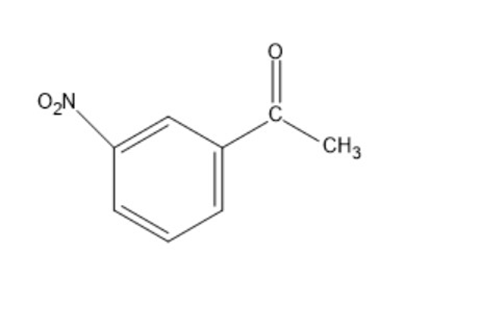
b)
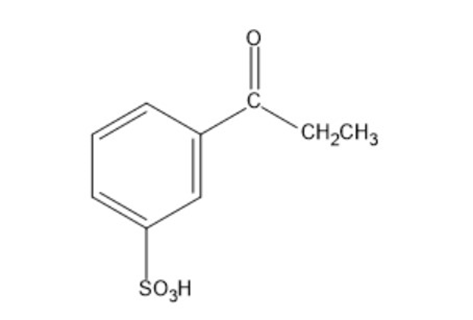
c)
a. Rank the following esters from most reactive to least reactive in the first slow step of a nucleophilic acyl substitution reaction (formation of the tetrahedral intermediate):
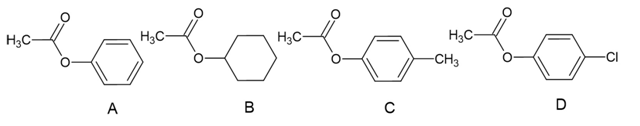
b. Rank the same esters from most reactive to least reactive in the second slow step of a nucleophilic acyl substitution reaction (collapse of the tetrahedral intermediate).
What do you think about this solution?
We value your feedback to improve our textbook solutions.