Chapter 18: Q51P (page 915)
Draw the product of each of the following reactions:
a.
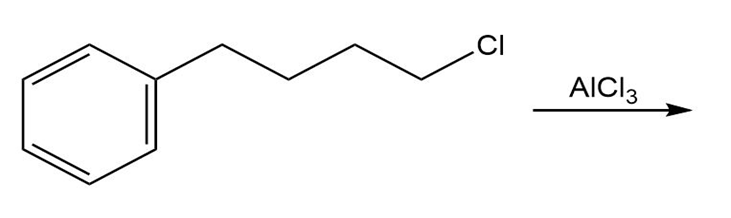
b.
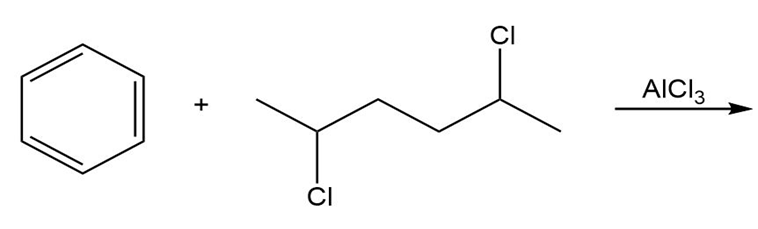
Short Answer
The product of each reactionsare,
a.
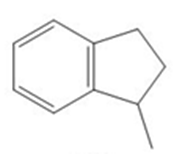
b.
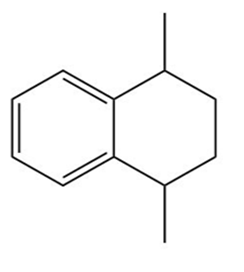
Learning Materials
Features
Discover
Chapter 18: Q51P (page 915)
Draw the product of each of the following reactions:
a.

b.

The product of each reactionsare,
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
A chemist isolated an aromatic compound with molecular formula C6H4Br2. He treated this compound with nitric acid and sulfuric acid and isolated three different isomers, in different amounts, with molecular formula C6H3Br2NO2. What was the structure of the original compound?
A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other.
Draw the structure of the activated benzene ring and the diazonium ion used in the synthesis of each of the following compounds, whose structures can be found on page 607.
a. butter yellow
b. methyl orange
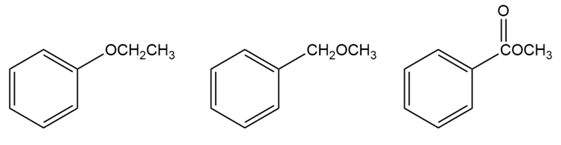
For each horizontal row of substituted benzenes, indicate
1.
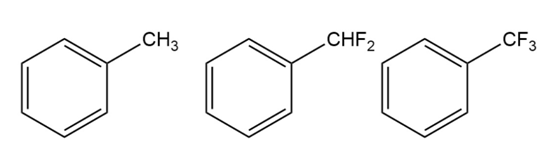
2.
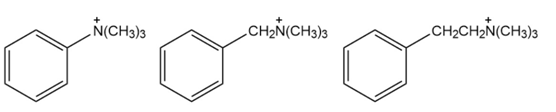
3.
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.