Chapter 16: Q63 P (page 793)
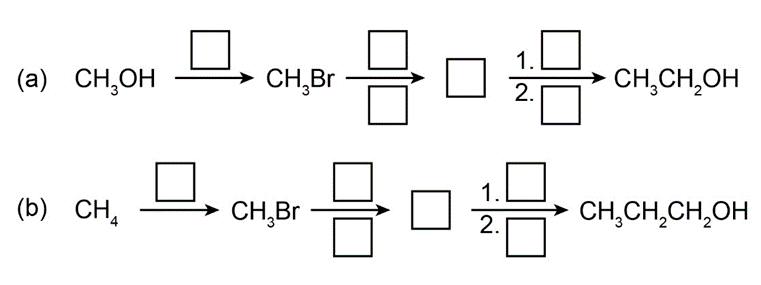
Question: Fill in the boxes:
Short Answer
Answer
a
b
Learning Materials
Features
Discover
Chapter 16: Q63 P (page 793)
Question: Fill in the boxes:

Answer
a
b
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the structure for each of the following:
a.
b.
c.
d.
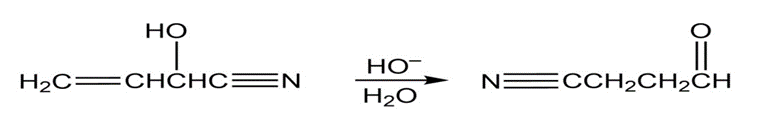
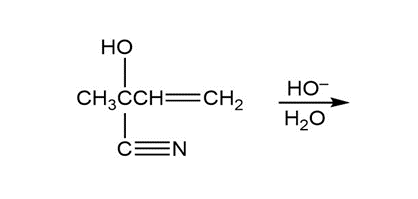
Question: Show two ways to convert an alkyl halide into a carboxylic acid that has one more carbon than the alkyl halide.
The pKa of protonated acetone is about -7.5, and the pKa of protonated hydroxylamine is 6.0.
a. In a reaction with hydroxylamine at pH 4.5 (Figure 16.2), what fraction of acetone is present in its acidic, protonated form? (Hint: See Section 2.10.)
b. In a reaction with hydroxylamine at pH 1.5, what fraction of acetone is present in its acidic, protonated form?
c. In a reaction with acetone at pH 1.5 (Figure 16.2), what fraction of hydroxylamine is present in its reactive basic form?
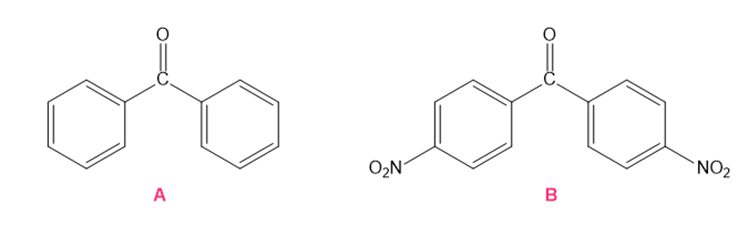
Which ketone forms the most hydrate in an aqueous solution?
What do you think about this solution?
We value your feedback to improve our textbook solutions.