Chapter 17: Q62P (page 848)
Show how the following compound can be synthesized from the given starting material.
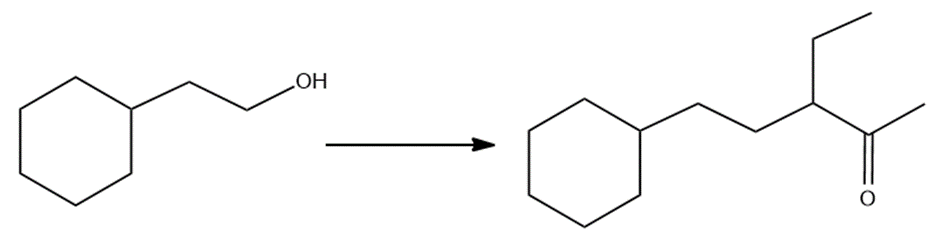
Short Answer
The answer is,
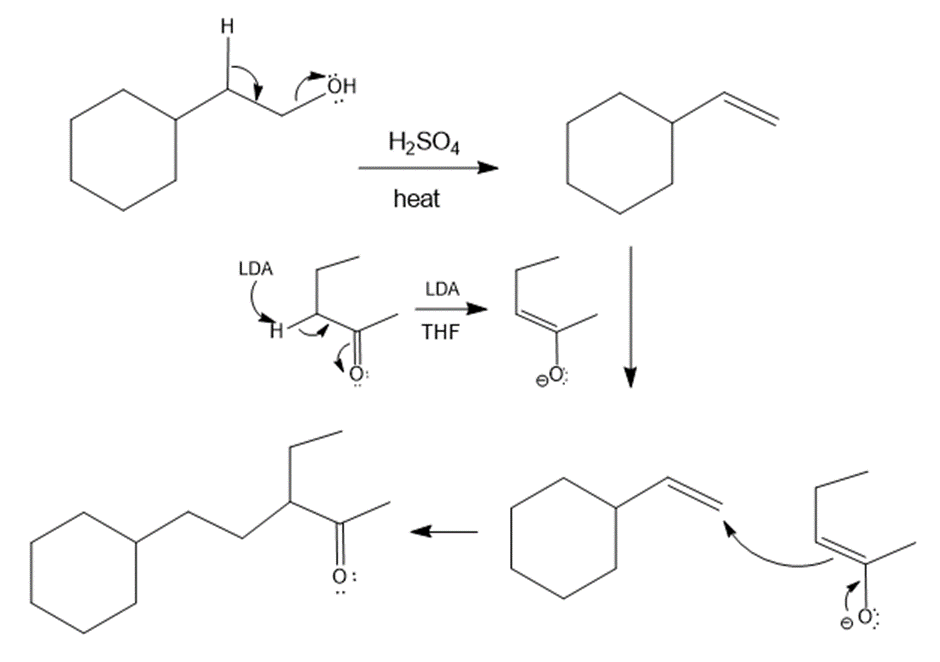
Reaction mechanism
Learning Materials
Features
Discover
Chapter 17: Q62P (page 848)
Show how the following compound can be synthesized from the given starting material.

The answer is,

Reaction mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
Problem: starting with bromocyclohexane, how can each of the following compounds be prepared?
The 1 H NMR chemical shifts of nitromethane, dinitromethane, and trinitromethane are at d 6.10, d 4.33, and d 7.52. Match each chemical shift with the compound. Explain how chemical shift correlates with pKa.
What two carbonyl compounds are required for the synthesis of morachalcone A (the aromatase inhibitor discussed in the box below), via a Claisen–Schmidt condensation?
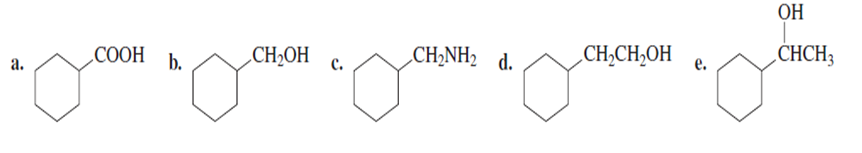
Which of the following compounds decarboxylates when heated?
a. What carboxylic acid is formed when the malonic ester synthesis is carried out with one equivalent of malonic ester, one equivalent of 1,5-dibromopentane, and two equivalents of base?
b. What carboxylic acid is formed when the malonic ester synthesis is carried out with two equivalents of malonic ester, one equivalent of 1,5-dibromopentane, and two equivalents of base?
What do you think about this solution?
We value your feedback to improve our textbook solutions.