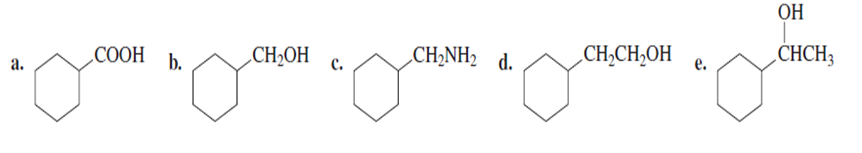
Chapter 17: 52P (page 847)
Which of the following compounds decarboxylates when heated?
Learning Materials
Features
Discover
Chapter 17: 52P (page 847)
Which of the following compounds decarboxylates when heated?
All the tools & learning materials you need for study success - in one app.
Get started for free
What product is obtained from the aldol condensation of cyclohexanone?
Propose a mechanism for the formation of fructose-1, 6-bisphosphate from dihydroxyacetone phosphate and glyceraldehyde-3-phosphate, using hydroxide ion as the catalyst.
Problem: starting with bromocyclohexane, how can each of the following compounds be prepared?
The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard reagent to add to the carbonyl group of an aldehyde or a ketone. Because the organozinc reagent is less reactive than a Grignard reagent, a nucleophilic addition to the ester group does not occur. The organozinc reagent is prepared by treating an ß-bromo ester with zinc.
Describe how each of the following compounds can be prepared, using a Reformatsky reaction:
A ketone undergoes acid-catalyzed bromination, acid-catalyzed chlorination, racemization, and acid-catalyzed deuterium exchange at the -carbon. All of these reactions have similar rate constants. What does this tell you about the mechanisms of these reactions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.