Chapter 17: Q20P (page 818)
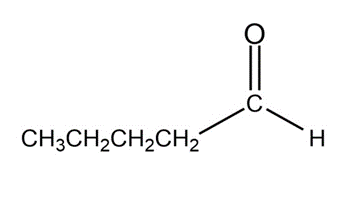
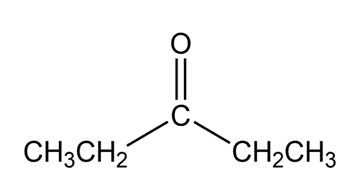
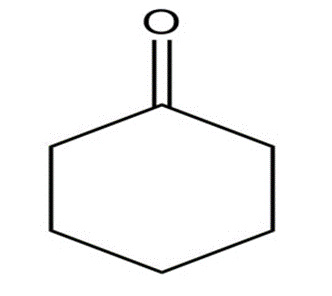
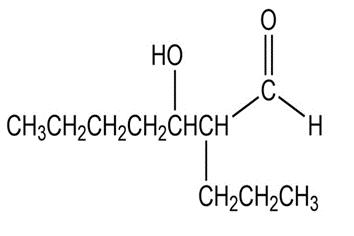
What aldol addition product is formed from each of the following compounds?
Learning Materials
Features
Discover
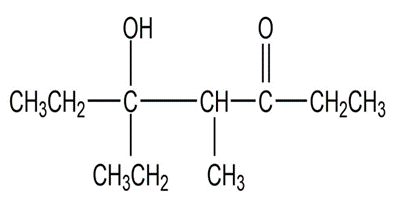
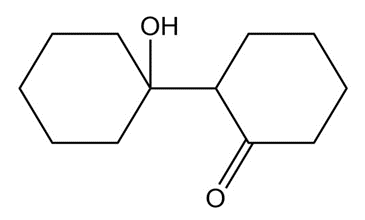
Chapter 17: Q20P (page 818)
What aldol addition product is formed from each of the following compounds?
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Explain why the -hydrogen of an N,N-disubstituted amide is less acidic (pKa= 30) than the hydrogen of an ester (pKa= 25).
Explain why the following carboxylic acids cannot be prepared by a malonic ester synthesis:
What reagents should be used to prepare the following compounds?
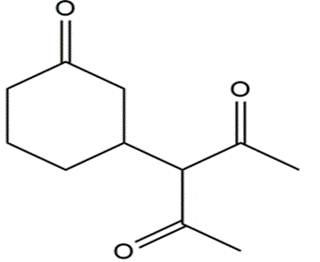
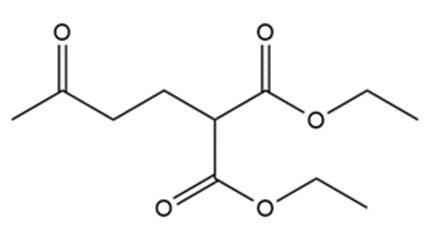
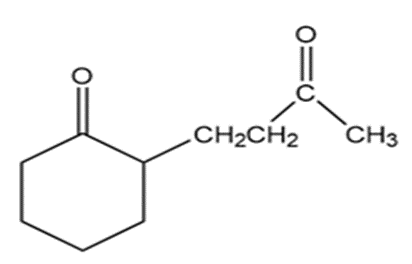
Show how the following compounds can be prepared from cyclohexanone:
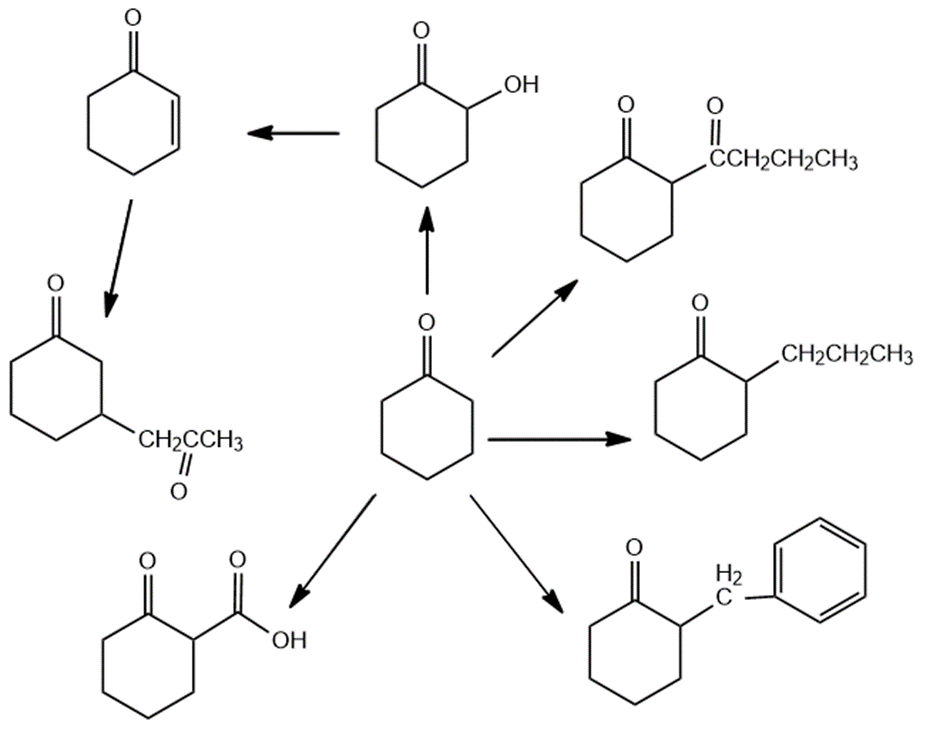
Do a retrosynthetic analysis on each of the following compounds, ending with the given starting materials:
(a)
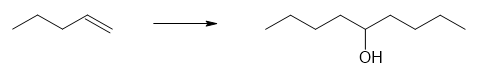
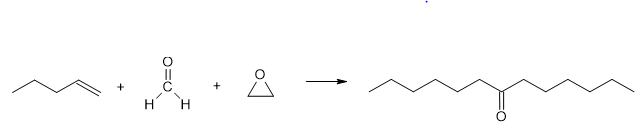
(b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.