Chapter 17: Q 6 TP (page 857)
Do a retrosynthetic analysis on each of the following compounds, ending with the given starting materials:
(a)
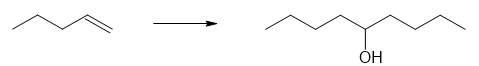
(b)
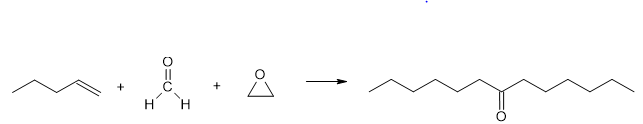
Short Answer
Answer:
(a)
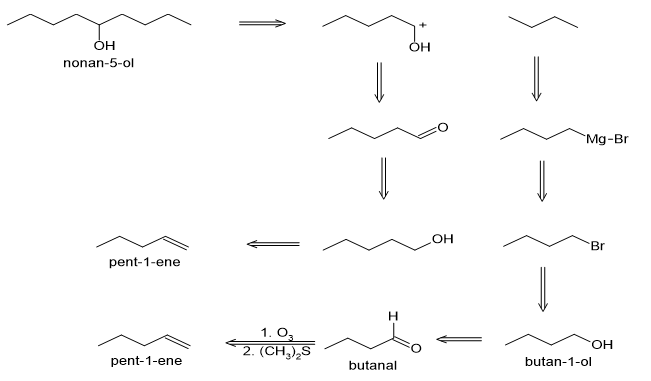
(b)
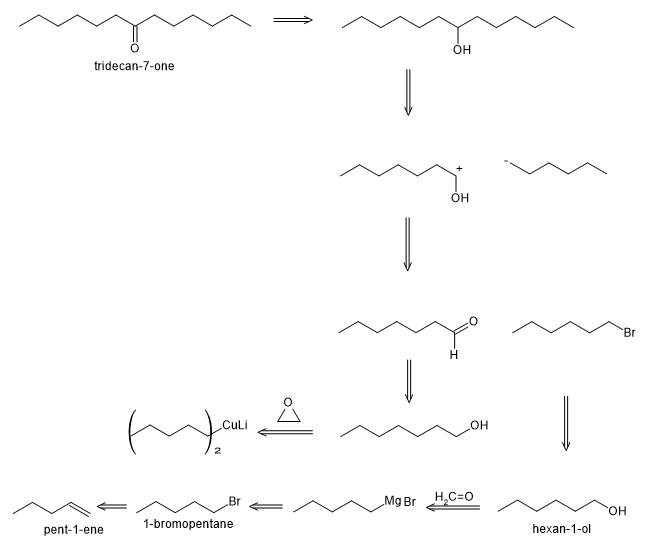
Learning Materials
Features
Discover
Chapter 17: Q 6 TP (page 857)
Do a retrosynthetic analysis on each of the following compounds, ending with the given starting materials:
(a)

(b)

Answer:
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
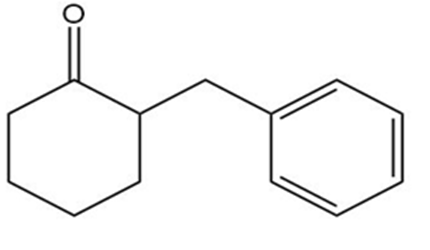
How could each of the following compounds be prepared from cyclohexanone?
a.
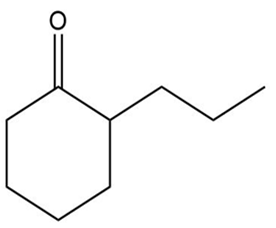
b.
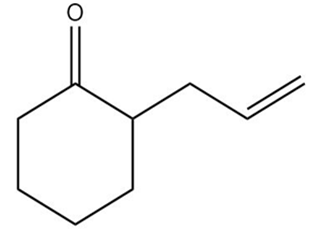
c.
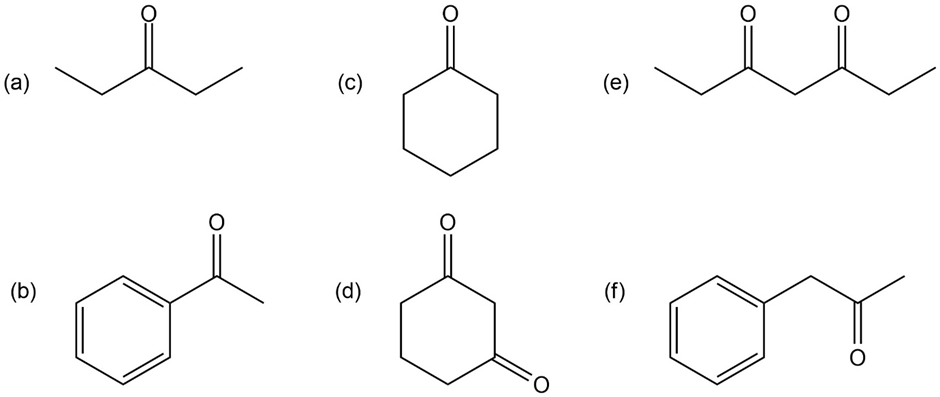
Draw the enol tautomer for each of the following compounds. For compounds that have more than one enol tautomer, indicate the one that is more stable.
Propose a mechanism for the following reaction:
Draw the products of the following reactions:
a.
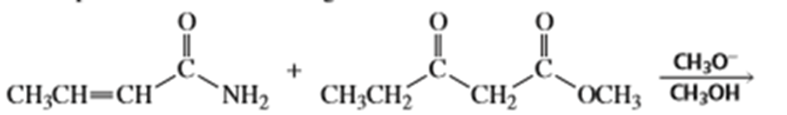
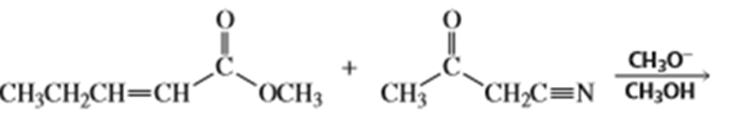
b.
Show how the following compounds can be synthesized. The only carbon-containing compounds available to you for each synthesis are shown.
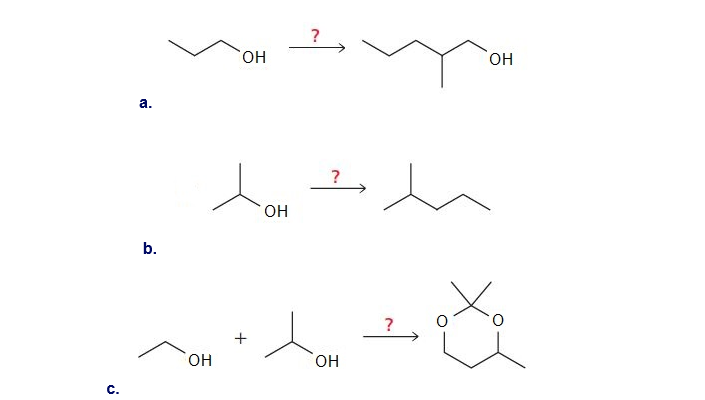
What do you think about this solution?
We value your feedback to improve our textbook solutions.