Chapter 17: Q-17-75P (page 850)
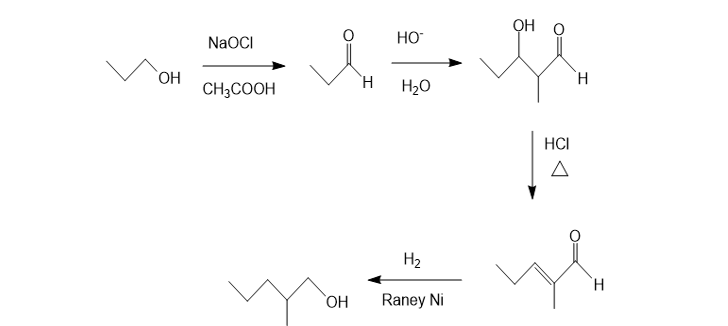
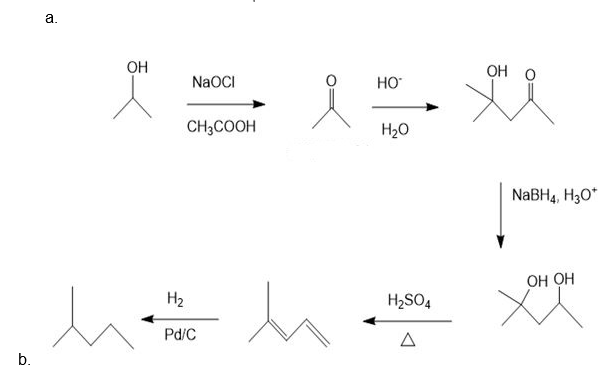
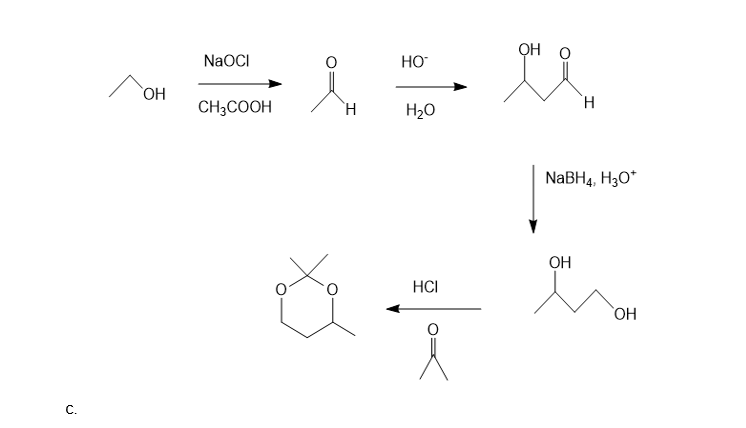
Show how the following compounds can be synthesized. The only carbon-containing compounds available to you for each synthesis are shown.
Short Answer
Learning Materials
Features
Discover
Chapter 17: Q-17-75P (page 850)
Show how the following compounds can be synthesized. The only carbon-containing compounds available to you for each synthesis are shown.




All the tools & learning materials you need for study success - in one app.
Get started for free
Describe three ways to synthesize the following compound:
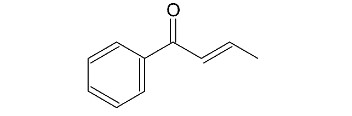
Propose a mechanism for the following reaction:
What two carbonyl compounds are required for the synthesis of morachalcone A (the aromatase inhibitor discussed in the box below), via a Claisen–Schmidt condensation?
The ketone whose1H NMR spectrum is shown here was obtained as the product of an acetoacetic ester synthesis. What alkyl halide was used in the synthesis?
When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl hydrogens are exchanged with deuterium but the hydrogen attached to the carbonyl carbon is not.
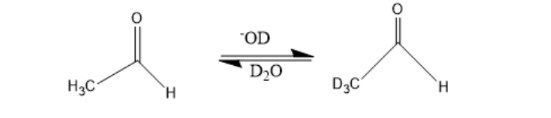
What do you think about this solution?
We value your feedback to improve our textbook solutions.