Chapter 17: Q16P (page 814)
How could each of the following compounds be prepared from cyclohexanone?
a.
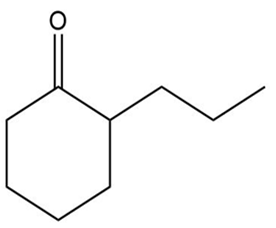
b.

c.

Short Answer
a.
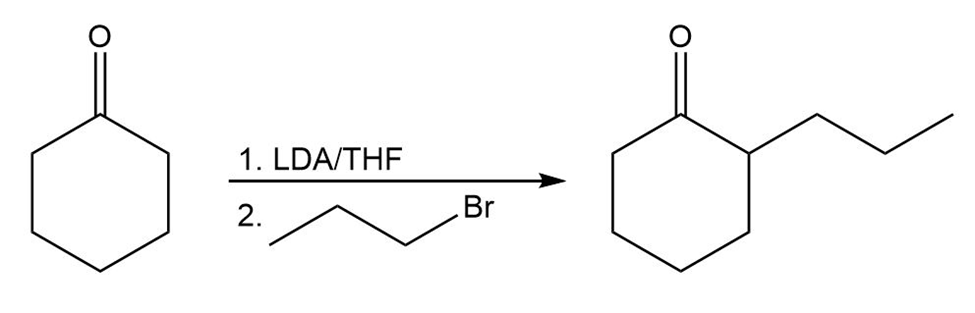
b.
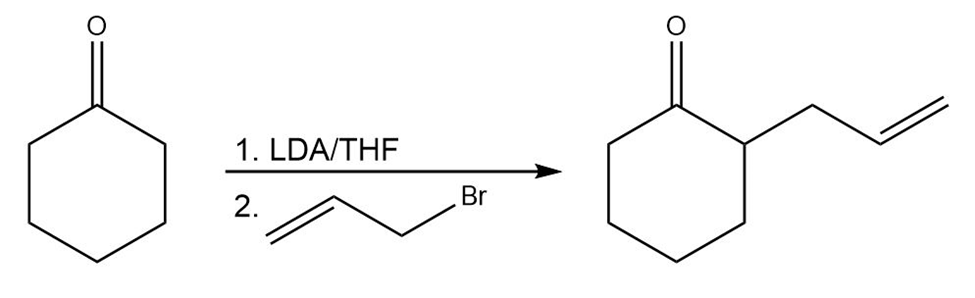
c.
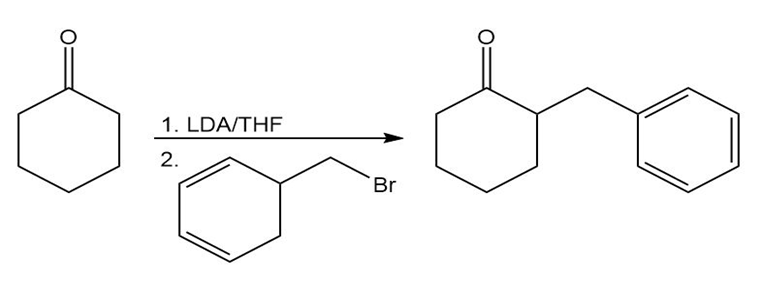
Learning Materials
Features
Discover
Chapter 17: Q16P (page 814)
How could each of the following compounds be prepared from cyclohexanone?
a.

b.

c.

a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
There are other condensation reactions similar to the aldol and claisen condensations:
a. The Perkin condensation is the condensation of an aromatic aldehyde and acetic anhydride. Draw the product obtained from the following Perkin condensation:

b. What compound is formed if water is added to the product of a Perkin condensation?
c. The Knoevenagel condensation is the condensation of an aldehyde or a ketone that has no alpha- hydrogens and a compound such as diethyl malonate that has an alpha-carbon flanked by two electron- withdrawing groups. Draw the product obtained from the following Knoevenagel condensation:
d. What product is obtained when the product of a Knoevenagel condensation is heated in an aqueous acidic solution?
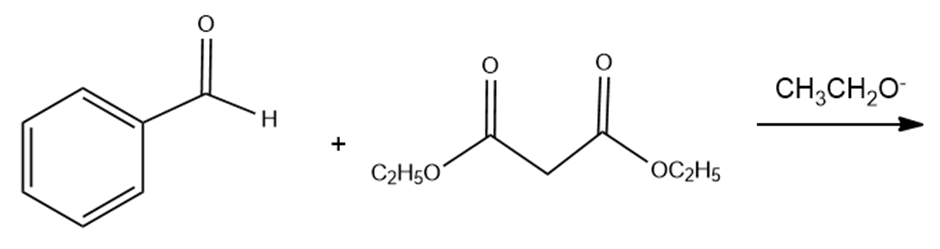
Draw the product of the reaction of each of the following compounds with a base:
Draw the products of the following reactions:
a. diethyl heptanedioate: (1) sodium ethoxide; (2) HCl
b. pentanoic acid + PBr3 + Br2, followed by hydrolysis
c. acetone + LDA/THF: (1) slow addition of ethyl acetate; (2) HCl
d. diethyl 2-ethylhexanedioate: (1) sodium ethoxide; (2) HCl
e. diethyl malonate: (1) sodium ethoxide; (2) isobutyl bromide; (3) HCl, H2O + ∆
f. acetophenone + LDA/THF: (1) slow addition of diethyl carbonate; (2) HCl
Draw the products of the following reactions:
a. 1,3-cyclohexanedione + LDA/THF, followed by allyl bromide
b. g-butyrolactone + LDA/THF, followed by methyl iodide
c. 2,7-octanedione + sodium hydroxide
d. diethyl 1,2-benzenedicarboxylate + sodium ethoxide: (1) slow addition of ethyl acetate; (2) HCl
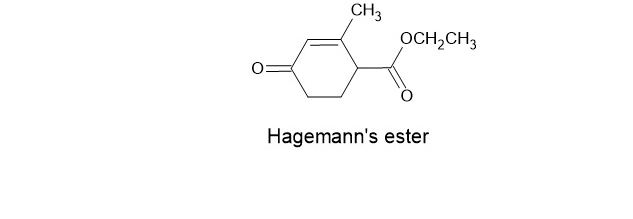
A compound known as Hagemann’s estercan be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid
and heat. Write the structure for the product of each of the steps.
a. The first step is an aldol-like condensation.
b. The second step is a Michael addition.
c. The third step is an intramolecular aldol addition.
d. The final transformation includes a dehydration and a hydrolysis followed by a decarboxylation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.