Chapter 28: Q44P (page 1239)
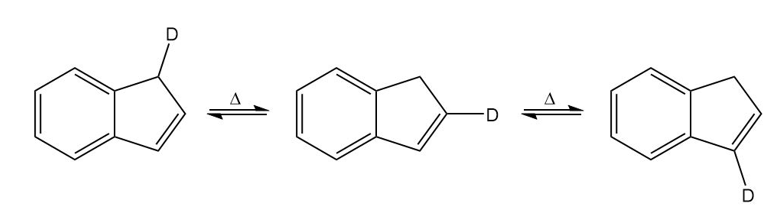
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.
Short Answer
Learning Materials
Features
Discover
Chapter 28: Q44P (page 1239)
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.


All the tools & learning materials you need for study success - in one app.
Get started for free
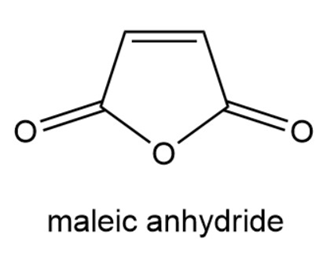
Explain why maleic anhydride reacts rapidly with 1,3-butadiene but does not react at all with ethene under thermal conditions.
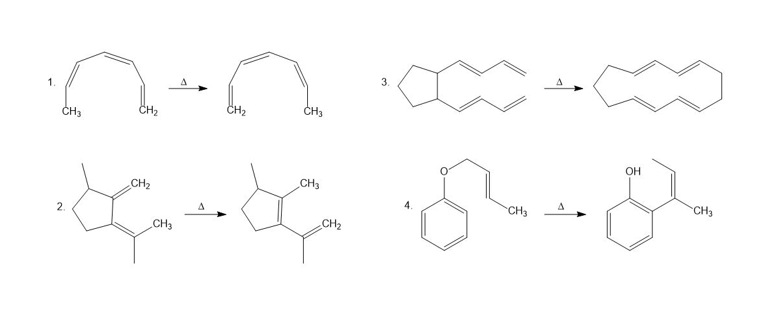
a. Name the kind of sigmatropic rearrangement that occurs in each of the following reactions.
b. Using arrows, show the electron rearrangement that takes place in each reaction.
Will a concerted reaction take place between 1,3-butadiene and 2-cyclohexenone in the presence of ultraviolet light?
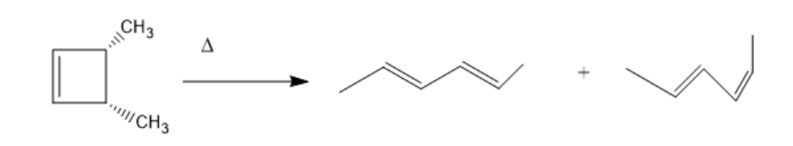
cis-3,4-Dimethylcyclobutene undergoes thermal ring opening to form the two products shown. One of the products is formed in 99% yield, the other in 1% yield. Which is which?
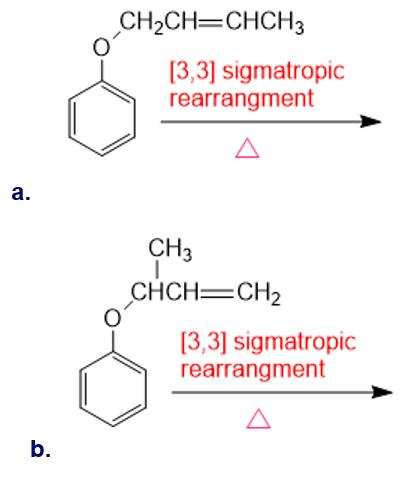
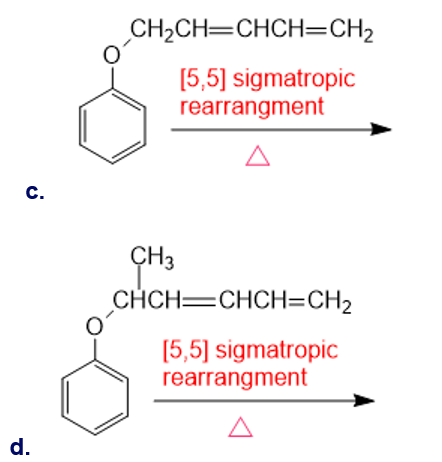
Draw the product of each of the following sigmatropic rearrangements:
What do you think about this solution?
We value your feedback to improve our textbook solutions.