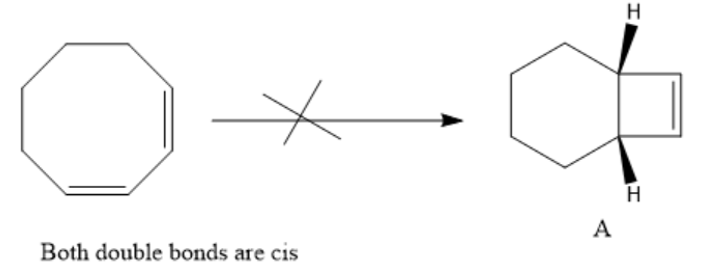
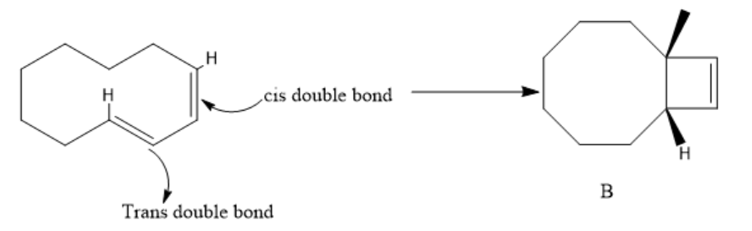
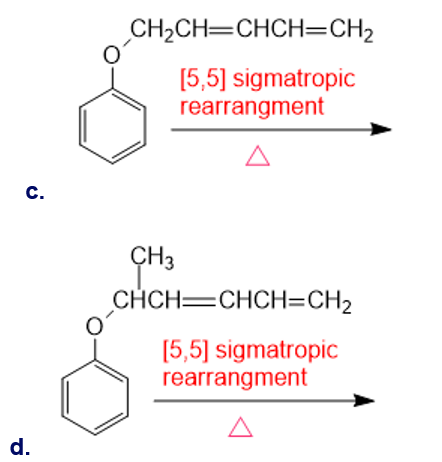
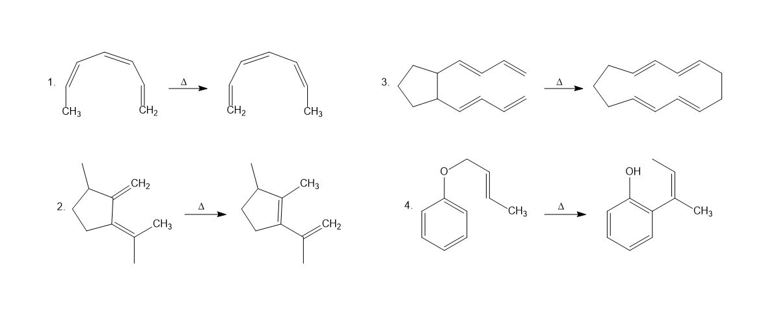
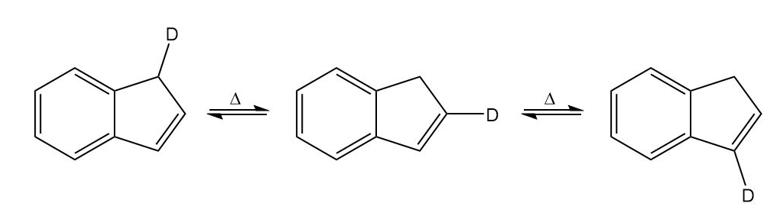
Chapter 28: Q43P (page 1239)
Explain why compound A will not undergo a ring-opening reaction under thermal conditions, but compound B will.
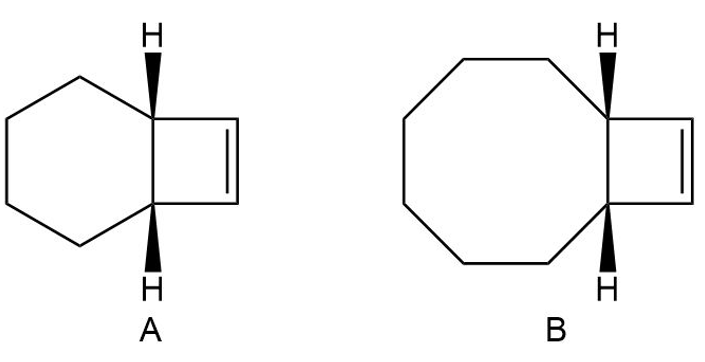
Short Answer
An eight-membered ring cannot accommodate a trans double bond in the ring.
A ten membered ring can accommodate a trans double bond in the ring.