Chapter 28: Q14P (page 1231)
Why was a deuterated compound used in the last reaction on the preceding page?
Short Answer
In the last reaction, a deuterated compound is used to detect the rearrangement type in the sigmatropic rearrangement reaction.
Learning Materials
Features
Discover
Chapter 28: Q14P (page 1231)
Why was a deuterated compound used in the last reaction on the preceding page?
In the last reaction, a deuterated compound is used to detect the rearrangement type in the sigmatropic rearrangement reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain why maleic anhydride reacts rapidly with 1,3-butadiene but does not react at all with ethene under thermal conditions.
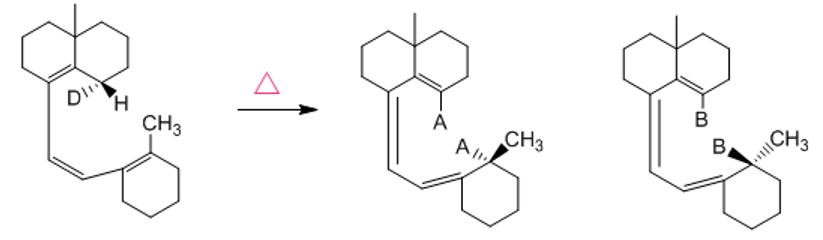
Two products are formed in the following [1,7] sigmatropic rearrangement, one due to hydrogen migration and the other to deuterium migration. Show the configuration of the products by replacing A and B with the appropriate atoms (H or D).
a. Propose a mechanism for the following reaction. (Hint:An electrocyclic reaction is followed by a Diels–Alder reaction.)
b. What would be the product if trans-2-butene were used instead of ethene?

Explain why two different products are formed from disrotatory ring closure of (2E,4Z,6Z)-octatriene, but only one product is formed from disrotatory ring closure of (2E,4Z,6E)-octatriene.
Will a concerted reaction take place between 1,3-butadiene and 2-cyclohexenone in the presence of ultraviolet light?
What do you think about this solution?
We value your feedback to improve our textbook solutions.