Chapter 28: Q11P (page 1227)
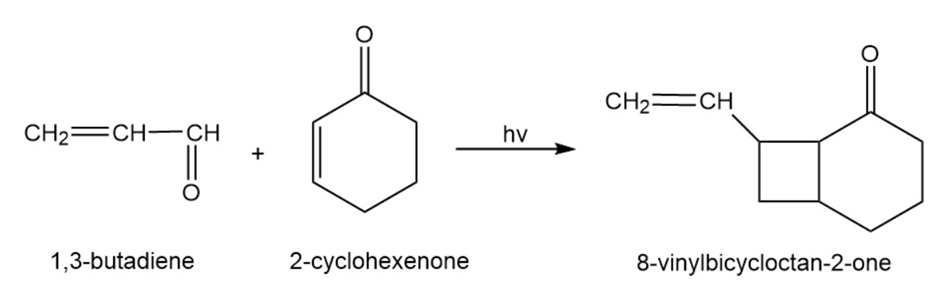
Will a concerted reaction take place between 1,3-butadiene and 2-cyclohexenone in the presence of ultraviolet light?
Short Answer
Learning Materials
Features
Discover
Chapter 28: Q11P (page 1227)
Will a concerted reaction take place between 1,3-butadiene and 2-cyclohexenone in the presence of ultraviolet light?

All the tools & learning materials you need for study success - in one app.
Get started for free
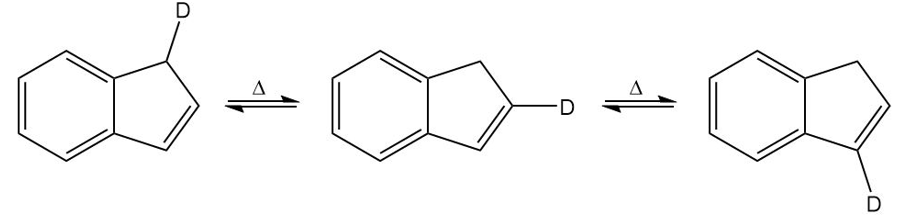
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.
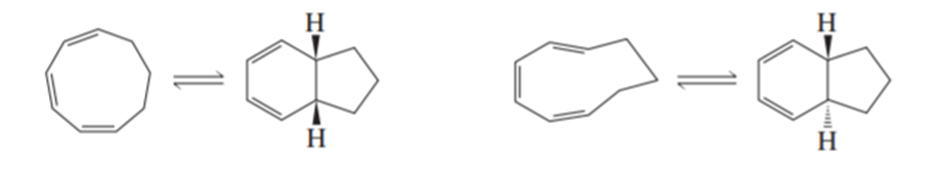
a. Propose a mechanism for the following reaction. (Hint:An electrocyclic reaction is followed by a Diels–Alder reaction.)
b. What would be the product if trans-2-butene were used instead of ethene?
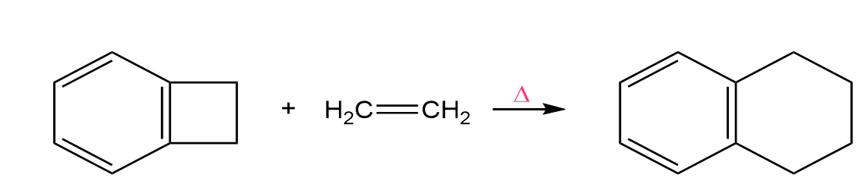
a. Draw the product of the following reaction:
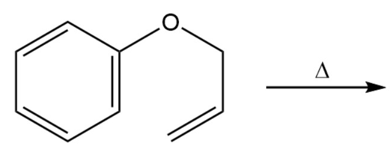
b. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.
Account for the difference in the products of the following reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.