Chapter 28: Q15P (page 1231)
Account for the difference in the products obtained under photochemical and thermal conditions:

Short Answer
a.
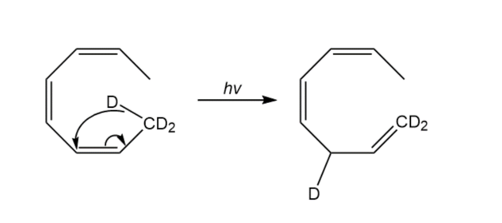
b.
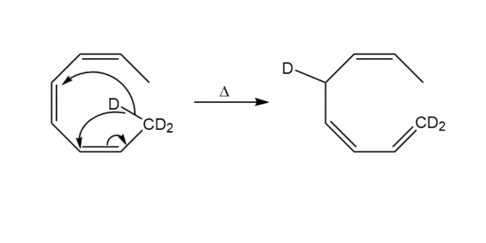
Learning Materials
Features
Discover
Chapter 28: Q15P (page 1231)
Account for the difference in the products obtained under photochemical and thermal conditions:

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism for the following reaction:
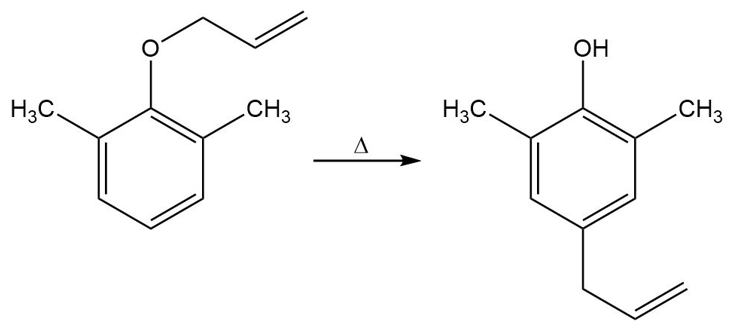
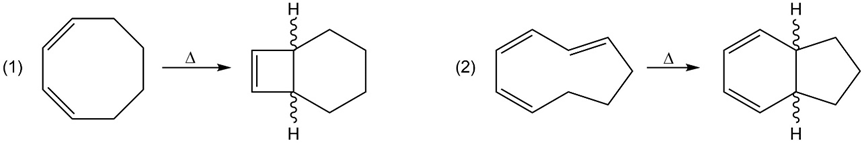
a. Identify the mode of ring closure for each of the following electrocyclic reactions.
b. Are the indicated hydrogens cis or trans?
Problem:Give a molecular orbital description for each of the following:
a. 1,3-pentadiene b. 1,4-pentadiene c. 1,3,5-heptatriene d. 1,3,5,8-nonatetraene
A student found that heating any one of the isomers shown here resulted in scrambling of the deuterium to all three positions on the five-membered ring. Propose a mechanism to account for this observation.
Problem:a. Under thermal conditions, will ring closure of (2E,4Z,6Z,8E)-2,4,6,8-decatetraene be conrotatory or disrotatory?
b. Will the product have the cis or the trans configuration?
c. Under photochemical conditions, will ring closure be conrotatory or disrotatory?
d. Will the product have the cis or the trans configuration?
What do you think about this solution?
We value your feedback to improve our textbook solutions.