Chapter 28: Q42P (page 1239)
Propose a mechanism for the following reaction:
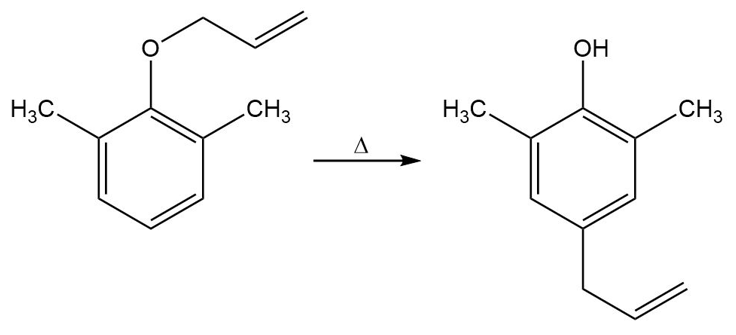
Short Answer
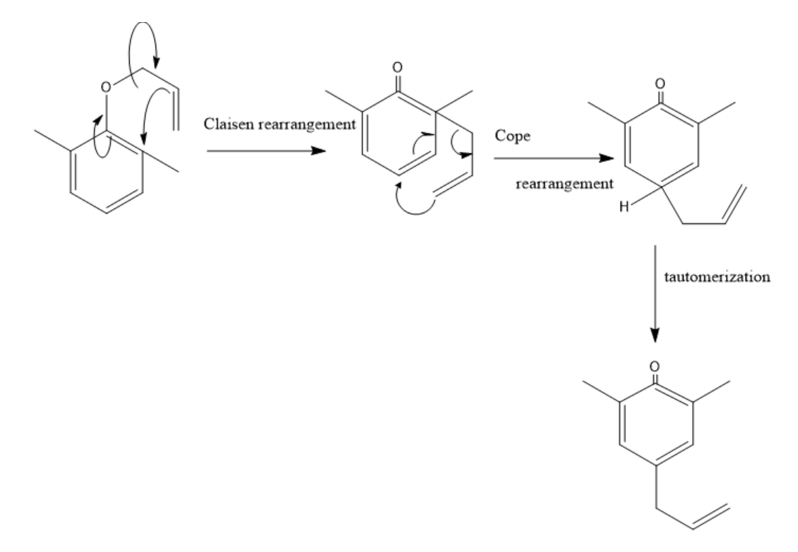
Learning Materials
Features
Discover
Chapter 28: Q42P (page 1239)
Propose a mechanism for the following reaction:


All the tools & learning materials you need for study success - in one app.
Get started for free
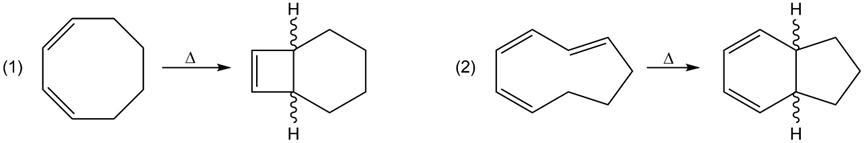
a. Identify the mode of ring closure for each of the following electrocyclic reactions.
b. Are the indicated hydrogens cis or trans?
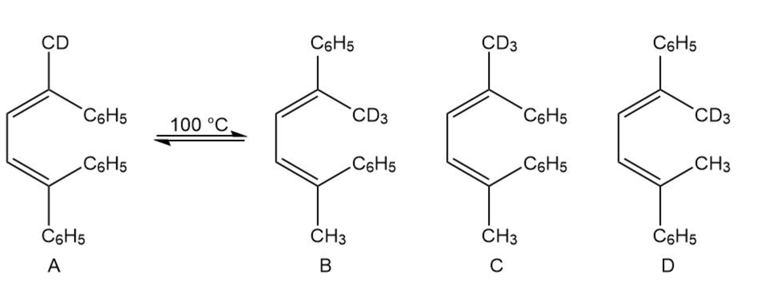
If isomer A is heated to about 100 °C, a mixture of isomers A and B is formed. Explain why there is no trace of isomer C or D.
Draw the product formed when each of the following compounds undergoes an electrocyclic reaction
a. under thermal conditions.
b. under photochemical conditions.

a. Draw the product of the following reaction:
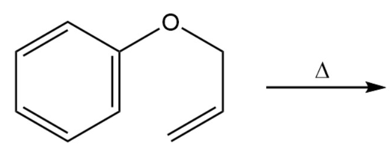
b. If the terminal sp2 carbon of the substituent attached to the benzene ring is labeled with 14C, where will the label be in the product?
Account for the difference in the products of the following reactions:

What do you think about this solution?
We value your feedback to improve our textbook solutions.