Chapter 28: Q40P (page 1239)
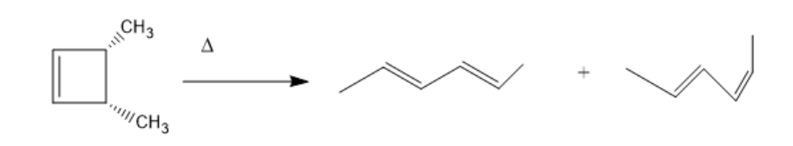
cis-3,4-Dimethylcyclobutene undergoes thermal ring opening to form the two products shown. One of the products is formed in 99% yield, the other in 1% yield. Which is which?
Short Answer

Learning Materials
Features
Discover
Chapter 28: Q40P (page 1239)
cis-3,4-Dimethylcyclobutene undergoes thermal ring opening to form the two products shown. One of the products is formed in 99% yield, the other in 1% yield. Which is which?


All the tools & learning materials you need for study success - in one app.
Get started for free
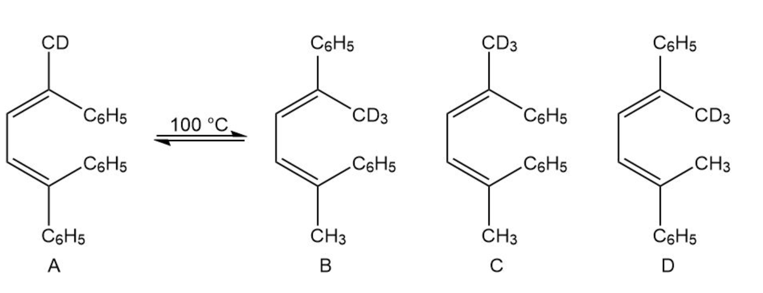
If isomer A is heated to about 100 °C, a mixture of isomers A and B is formed. Explain why there is no trace of isomer C or D.
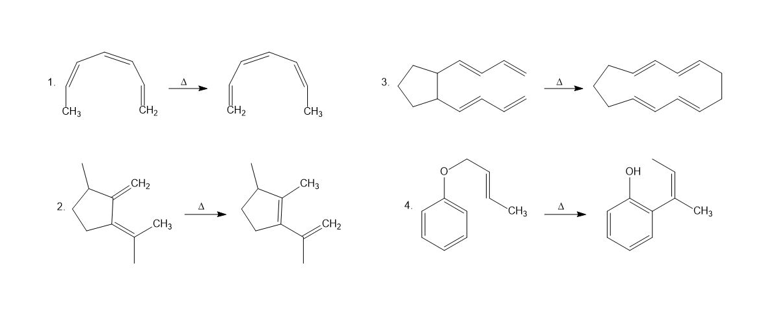
a. Name the kind of sigmatropic rearrangement that occurs in each of the following reactions.
b. Using arrows, show the electron rearrangement that takes place in each reaction.
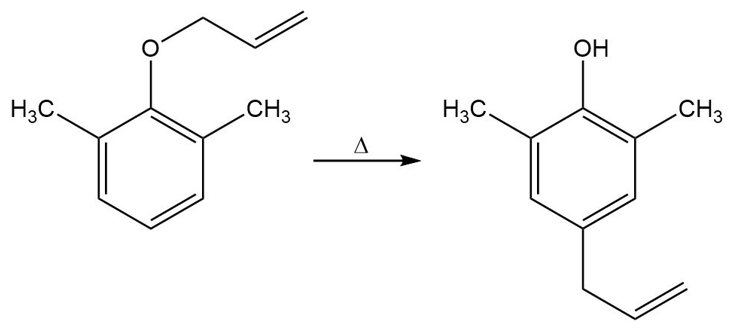
Propose a mechanism for the following reaction:
Problem:Which of the following are correct? Correct any false statements.
a. A conjugated diene with an even number of double bonds undergoes conrotatory ring closure under thermal conditions.
b. A conjugated diene with an antisymmetric HOMO undergoes conrotatory ring closure under thermal conditions.
c. A conjugated diene with an odd number of double bonds has a symmetric HOMO.
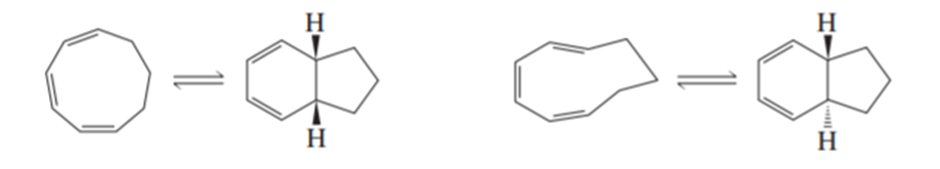
Account for the difference in the products of the following reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.