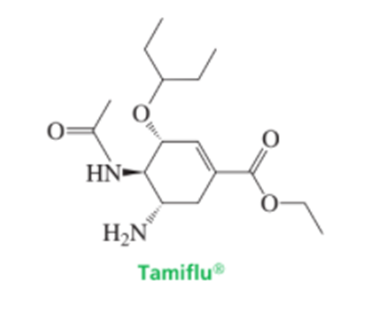
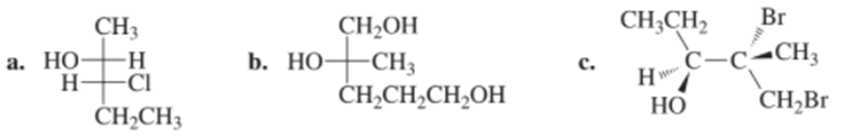Chapter 4: Q95P (page 185)
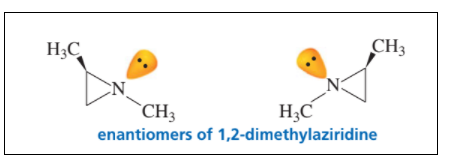
Explain why the enantiomers of 1,2-dimethylaziridine can be separated even though one of the “groups” attached to nitrogen is a lone pair.
Short Answer
In this compound nitrogen is in sp2 hybridization. But it don’t have an angle of 120°. In the transition state of amine inversion the nitrogen atom is sp2 hybridized so the bond angle is 120°. A nitrogen is three membered ring cannot achieve a bond angle of 120°. So the amine inversion that would interconvert the enantiomers cannot occur. Therefore the enantiomers can’t be separated.