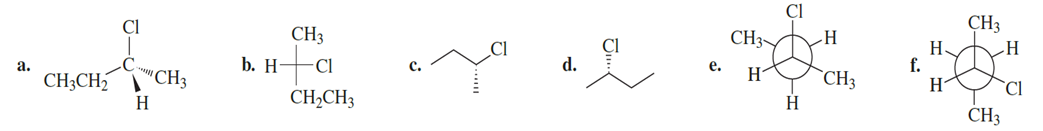
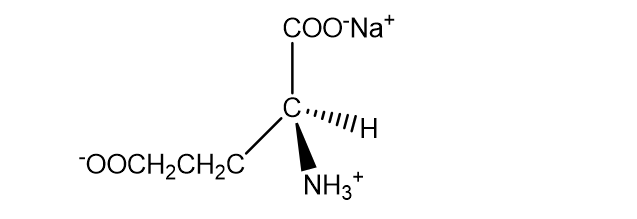
Chapter 4: Q96P (page 185)
A sample of (S)-(+)-lactic acid was found to have an enantiomeric excess of 72%. How much Risomer is present in the sample?
Short Answer
The actual amount of S-isomer present in the sample is = 72% +  =86%.
=86%.
Hence the amount of R-isomer present in the sample is = 100% - 86% = 14%.

 = 86%
= 86%