Chapter 3: Q19P (page 101)
Draw a condensed and a skeletal structure for each of the following:
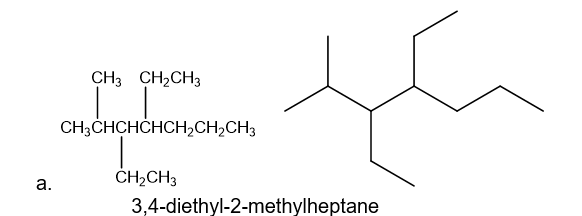
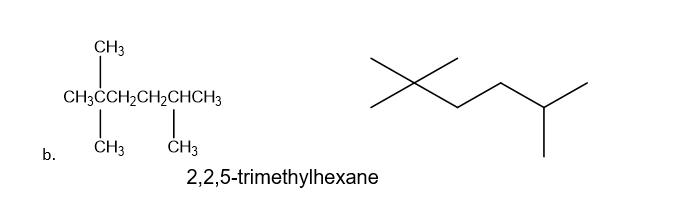
a. 3,4-diethyl-2-methylheptane
b. 2,2,5-trimethylhexane
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q19P (page 101)
Draw a condensed and a skeletal structure for each of the following:
a. 3,4-diethyl-2-methylheptane
b. 2,2,5-trimethylhexane


All the tools & learning materials you need for study success - in one app.
Get started for free
Which of the following represents a cis isomer?
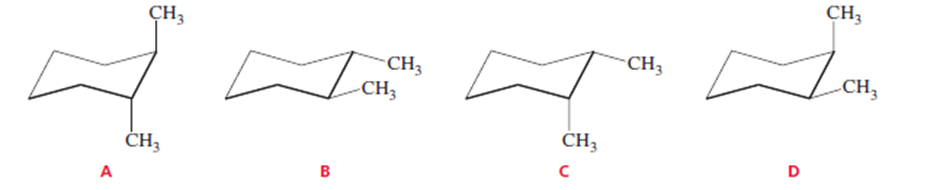
One of the chair conformers of cis-1,3-dimethylcyclohexane is 5.4 kcal/mol less stable than the other. How much steric strain does a 1,3-diaxial interaction between two methyl groups introduce into the conformer?
Draw the structures and provide systematic names for parts a, b, and c by substituting a chlorine for a hydrogen of methylcyclohexane:
a. A primary alkyl halide
b. A tertiary alkyl halide
c. Three secondary alkyl halides
Explain why
a. H2O (100 °C) has a higher boiling point than CH3OH (65 °C)
b. H2O (100 °C) has a higher boiling point than NH3 (-33 °C).
c. H2O (100 °C) has a higher boiling point than HF (20 °C).
d. HF (20 °C) has a higher boiling point than NH3 (-33 °C).
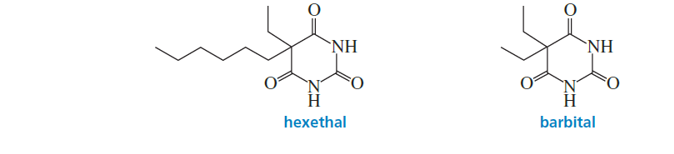
The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the nonpolar membrane of a cell. Which of the following barbiturates would you expect to be the more effective sedative?
What do you think about this solution?
We value your feedback to improve our textbook solutions.