Chapter 3: Q58 P (page 137)
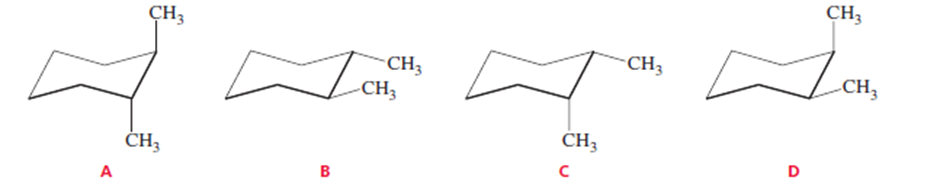
Which of the following represents a cis isomer?
Short Answer
Compounds C and D represent cis isomers.
Learning Materials
Features
Discover
Chapter 3: Q58 P (page 137)
Which of the following represents a cis isomer?

Compounds C and D represent cis isomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Using the data obtained in Problem 81, calculate the percentage of molecules of trans-1,2-dimethylcyclohexane that will have both methyl groups in
equatorial positions.
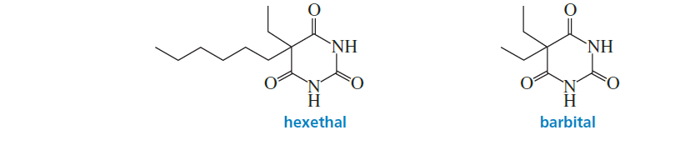
The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the nonpolar membrane of a cell. Which of the following barbiturates would you expect to be the more effective sedative?
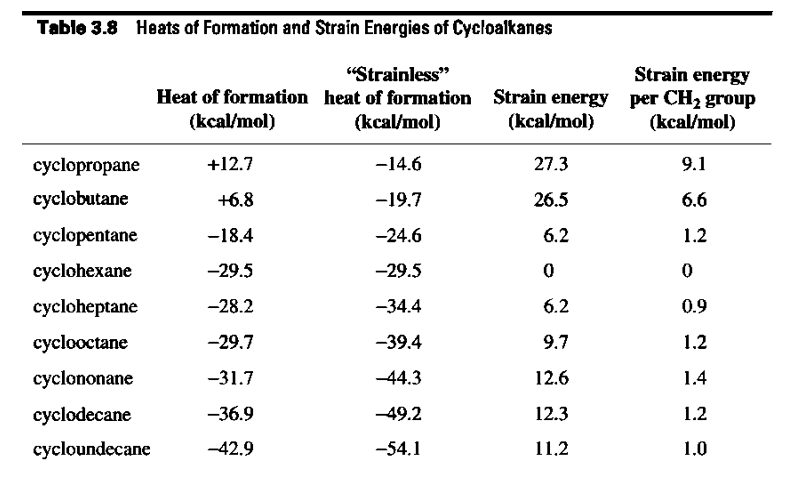
Verify the strain energy shown in Table 3.8 for cycloheptane.
Give each of the following a systematic name and indicate whether each is a primary, secondary, or tertiary alcohol:

Draw a picture of the hydrogen bonding in methanol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.