Chapter 3: Q.3-39P (page 118)
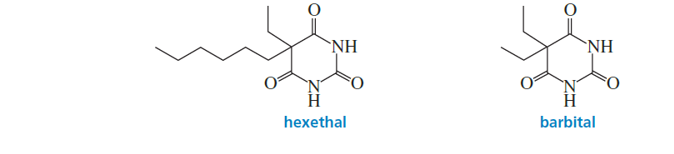
The effectiveness of a barbiturate as a sedative is related to its ability to penetrate the nonpolar membrane of a cell. Which of the following barbiturates would you expect to be the more effective sedative?
Short Answer
The hexethal will be a more effective sedative because of the presence of a longer hydrophobic chain than barbital.

