Chapter 3: Q.3-44P (page 124)
Verify the strain energy shown in Table 3.8 for cycloheptane.

Short Answer
Strain energy of cycloheptane = actual heat of formation - strain less heat of formation
= 6.2 Kcal/mol
Learning Materials
Features
Discover
Chapter 3: Q.3-44P (page 124)
Verify the strain energy shown in Table 3.8 for cycloheptane.

Strain energy of cycloheptane = actual heat of formation - strain less heat of formation
= 6.2 Kcal/mol
All the tools & learning materials you need for study success - in one app.
Get started for free
Which has
a. the higher boiling point: 1-bromopentane or 1-bromohexane?
b. the higher boiling point: pentyl chloride or isopentyl chloride?
c. the greater solubility in water: 1-butanol or 1-pentanol?
d. the higher boiling point: 1-hexanol or 1-methoxypentane?
e. the higher melting point: hexane or isohexane?
f. the higher boiling point: 1-chloropentane or 1-pentanol?
g. the higher boiling point: 1-bromopentane or 1-chloropentane?
h. the higher boiling point: diethyl ether or butyl alcohol?
i. the greater density: heptane or octane?
j. the higher boiling point: isopentyl alcohol or isopentylamine?
k. the higher boiling point: hexylamine or dipropylamine?
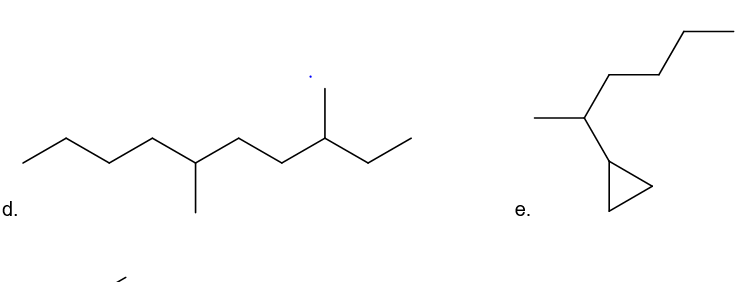
What is each compound’s systematic name?


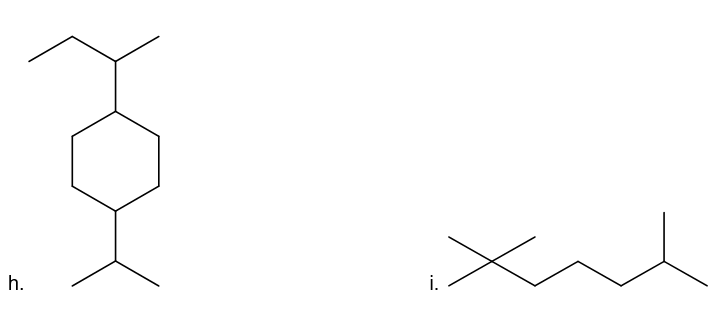
For each of the following, give the systematic name and the common name (if it has one) and then indicate whether it is a primary, secondary, or tertiary amine.

a. Which of the following compounds forms hydrogen bonds between its molecules?
1. CH3CH2OCH2CH2OH 3. CH3CH2CH2CH2Br 5. CH3CH2CH2COOH
2. CH3CH2N(CH3)2 4. CH3CH2CH2NHCH3 6. CH3CH2CH2CH2F
b. Which of the preceding compounds forms hydrogen bonds with a solvent such as ethanol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.