Chapter 3: Q18P (page 101)
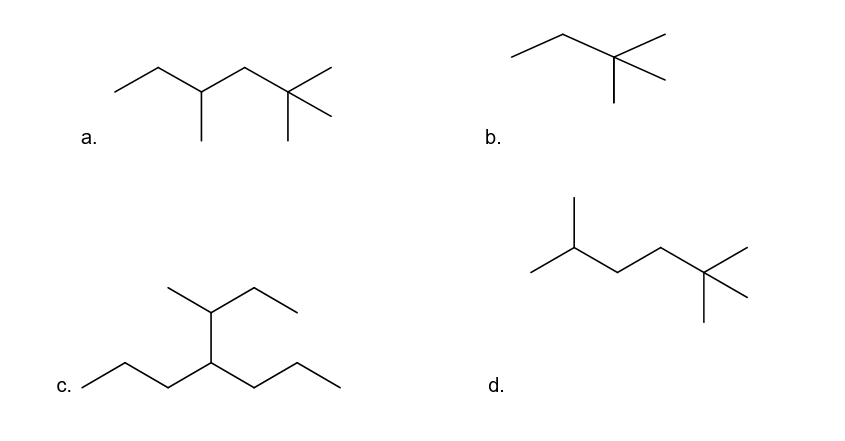
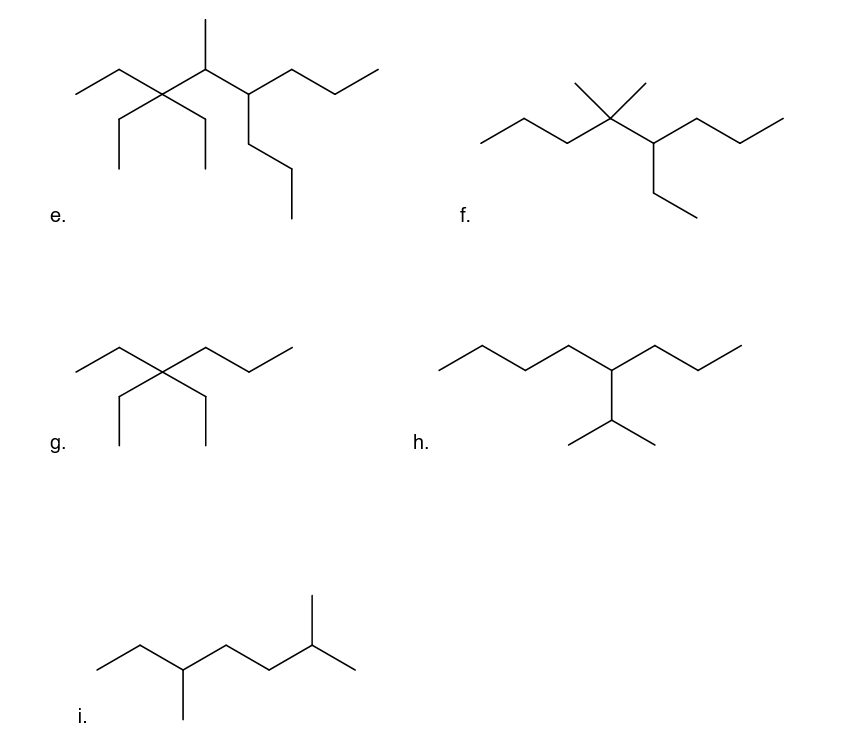
Convert the structures in Problem 9 into skeletal structures.
Structure in Problem 9
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q18P (page 101)
Convert the structures in Problem 9 into skeletal structures.
Structure in Problem 9



All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure for each of the following:
a. 2,2-dimethyl-4-isopropyloctane
b. 2,3-dimethylhexane
c. 4,4-diethyldecane
d. 2,4,5-trimethyl-4-(1-methylethyl) heptane
e. 2,5-dimethyl-4-(2-methylpropyl) octane
f. 4-(1,1-dimethylethyl) octane
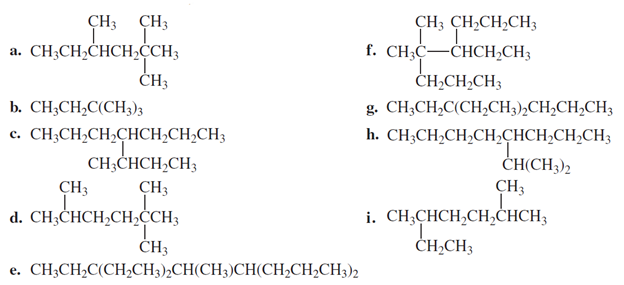
Name the following compounds:
For rotation about the C-3 -C-4 bond of 2-methylhexane, do the following: a. Draw the Newman projection of the most stable conformer.
b. Draw the Newman projection of the least stable conformer.
c. About which other carbon–carbon bonds may rotation occur?
d. How many of the carbon–carbon bonds in the compound have staggered conformers that are all equally stable?
Draw a condensed and a skeletal structure for each of the following:
a. 3,4-diethyl-2-methylheptane
b. 2,2,5-trimethylhexane
Draw the structures of a homologous series of alcohols that have from one to six carbons and give each of them a common name and a systematic name.
What do you think about this solution?
We value your feedback to improve our textbook solutions.