Chapter 3: Q17P (page 101)
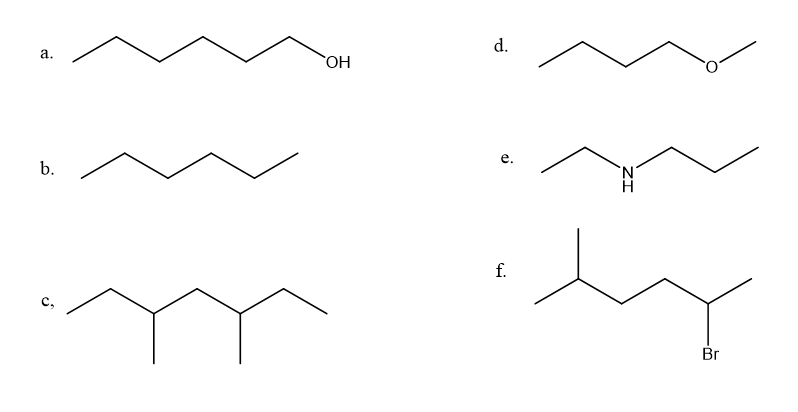
Convert the following condensed structures into skeletal structures:
a. CH3CH2CH2CH2CH2CH2OH
b. CH3CH2CH2CH2CH2CH3
c.CH3CH2CHCH2CHCH2CH3CH3CH3
d. CH3CH2CH2CH2OCH3
e. CH3CH2NHCH2CH2CH3
f.CH3CH2NHCH2CH2CH3
Short Answer
Learning Materials
Features
Discover
Chapter 3: Q17P (page 101)
Convert the following condensed structures into skeletal structures:
a. CH3CH2CH2CH2CH2CH2OH
b. CH3CH2CH2CH2CH2CH3
c.CH3CH2CHCH2CHCH2CH3CH3CH3
d. CH3CH2CH2CH2OCH3
e. CH3CH2NHCH2CH2CH3
f.CH3CH2NHCH2CH2CH3

All the tools & learning materials you need for study success - in one app.
Get started for free
A student was given the structural formulas of several compounds and was asked to give them systematic names. How many did the student name correctly? Correct those that are misnamed.
l. 2-methyl-N, N-dimethyl-4-hexanamine
The bond angles in a regular polygon with nsides are equal to
a. What are the bond angles in a regular octagon?
b. What are the bond angles in a regular nonagon?
a. Which of the following compounds forms hydrogen bonds between its molecules?
1. CH3CH2OCH2CH2OH 3. CH3CH2CH2CH2Br 5. CH3CH2CH2COOH
2. CH3CH2N(CH3)2 4. CH3CH2CH2NHCH3 6. CH3CH2CH2CH2F
b. Which of the preceding compounds forms hydrogen bonds with a solvent such as ethanol?
For rotation about the C-3 -C-4 bond of 2-methylhexane, do the following: a. Draw the Newman projection of the most stable conformer.
b. Draw the Newman projection of the least stable conformer.
c. About which other carbon–carbon bonds may rotation occur?
d. How many of the carbon–carbon bonds in the compound have staggered conformers that are all equally stable?
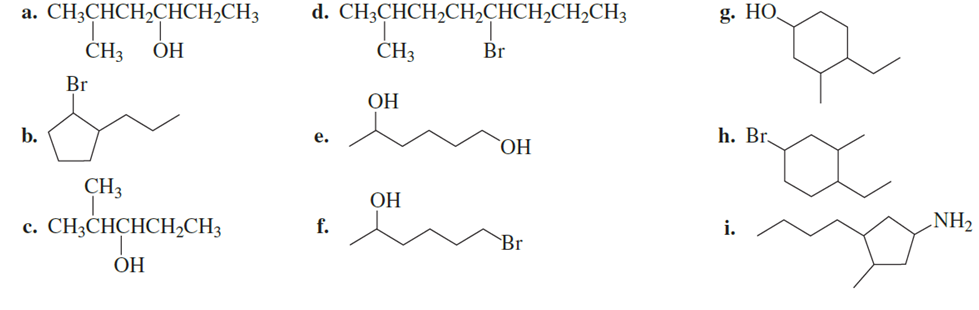
What is each compound’s systematic name?
What do you think about this solution?
We value your feedback to improve our textbook solutions.