Chapter 21: Q37P (page 1014)
Write the mechanism for the reaction of a cysteine side chain with iodoacetic acid.
Short Answer
The answer is:
 Mechanism
Mechanism
Learning Materials
Features
Discover
Chapter 21: Q37P (page 1014)
Write the mechanism for the reaction of a cysteine side chain with iodoacetic acid.
The answer is:
 Mechanism
Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the difference in the \({\bf{p}}{{\bf{K}}_{\bf{a}}}\) values of the carboxyl groups of alanine, serine, and cysteine.
Alanine has pKa values of 2.34 and 9.69. Therefore, alanine exists predominately as a zwitterion in an aqueous solution with pH > ____ and pH < ____.
What aldehyde is formed when valine is treated with ninhydrin?
Draw the product obtained when a lysine side chain in a polypeptide reacts with maleic anhydride.
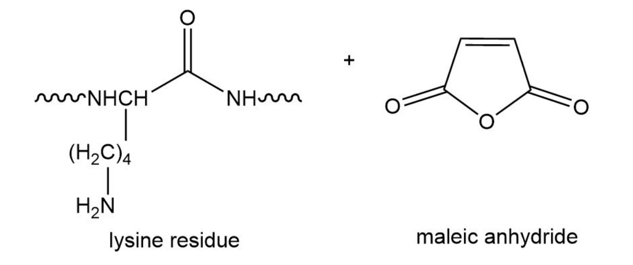
In what order would histidine, serine, aspartate, and valine be eluted with a buffer of pH 4 from a columncontaining an anion-exchange resin (Dowex 1)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.