Chapter 21: Q36P (page 1012)
Show the steps in the synthesis of the tetrapeptide in problem 34, using Merrifield’s method.
Short Answer
The answer is,
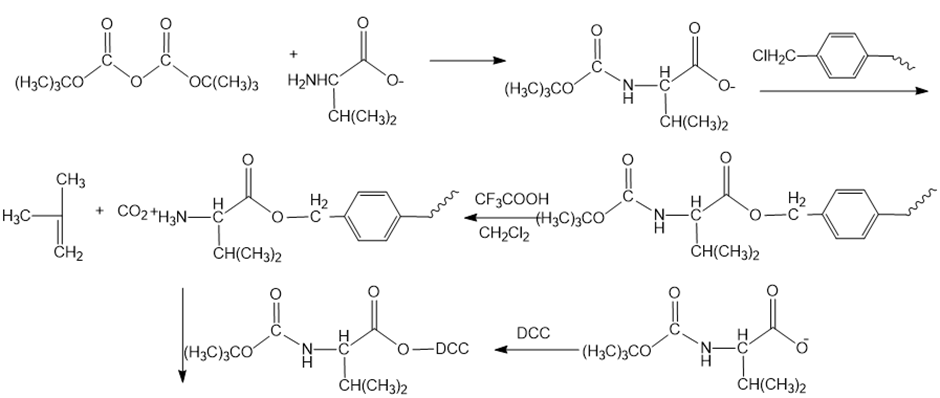
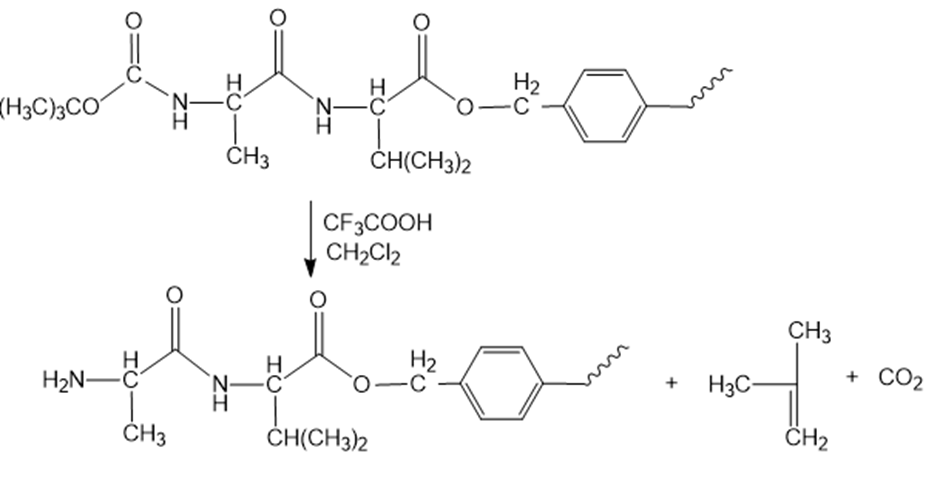
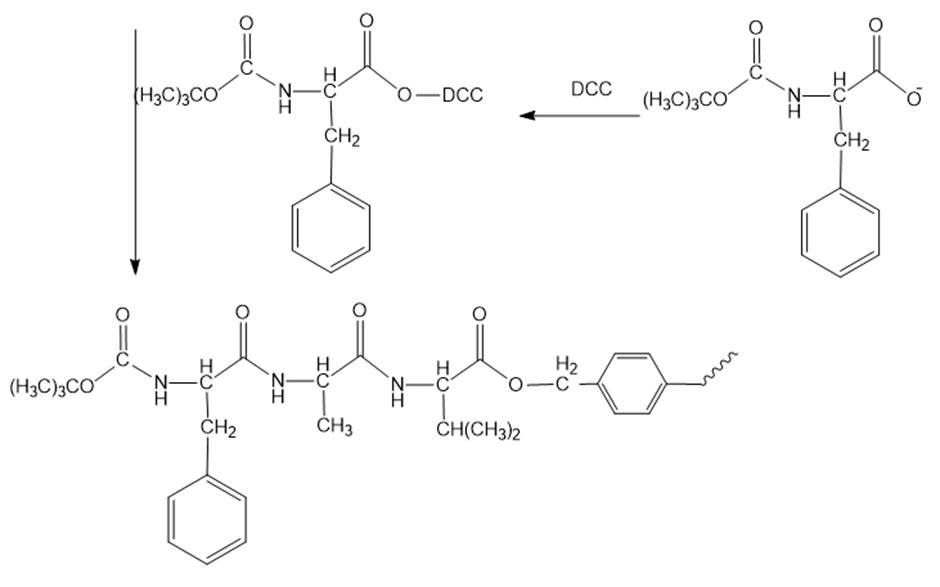
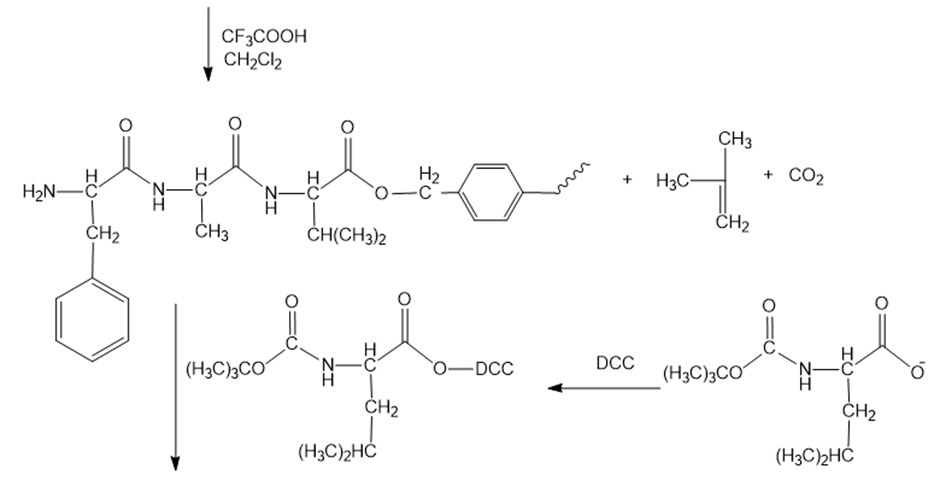
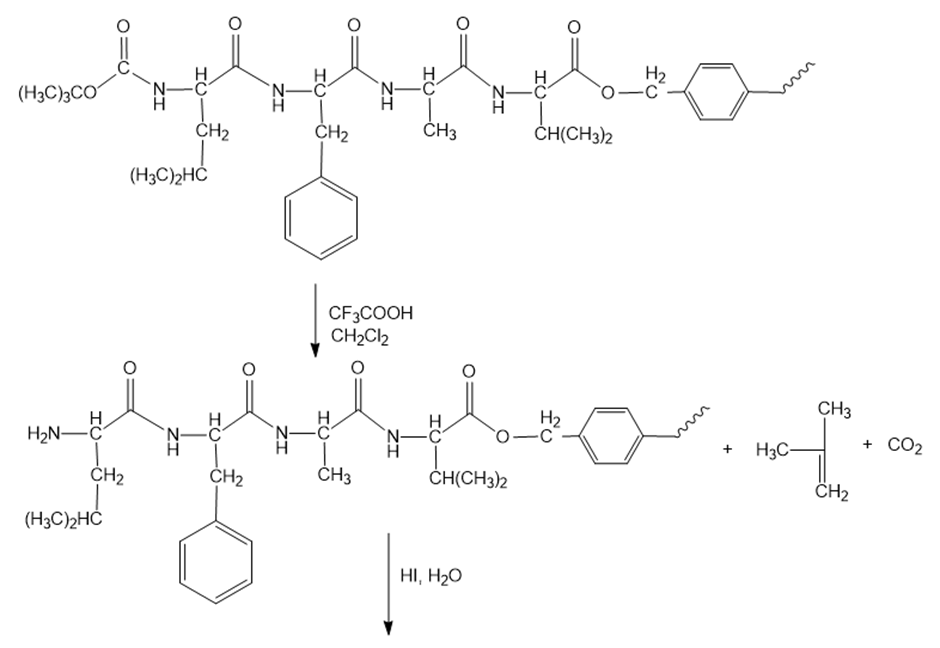
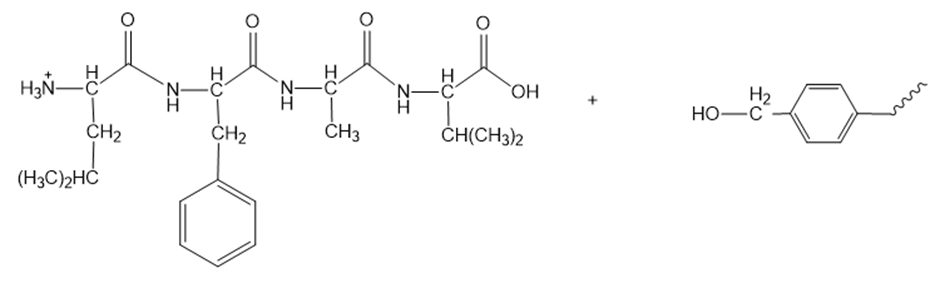
Reaction
Learning Materials
Features
Discover
Chapter 21: Q36P (page 1012)
Show the steps in the synthesis of the tetrapeptide in problem 34, using Merrifield’s method.
The answer is,






Reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how aspartame can be synthesized using DCCD.
Question: In determining the primary structure of insulin, what would lead you to conclude that insulin had more than one polypeptide chain?
Glutathione is a tripeptide whose function is to destroy harmful oxidizing agents in the body. Oxidizing agents are bought to be responsible for some of the effects of aging and to play a causative role in cancer.
Glutathione removes oxidizing agents by reducing them. In the process, glutathione is oxidized, resulting in the formation of a disulphide bond between two glutathione molecules. An enzyme subsequently reduces the disulphide bond, returning glutathione to its original condition so it can react with another oxidizing agent.
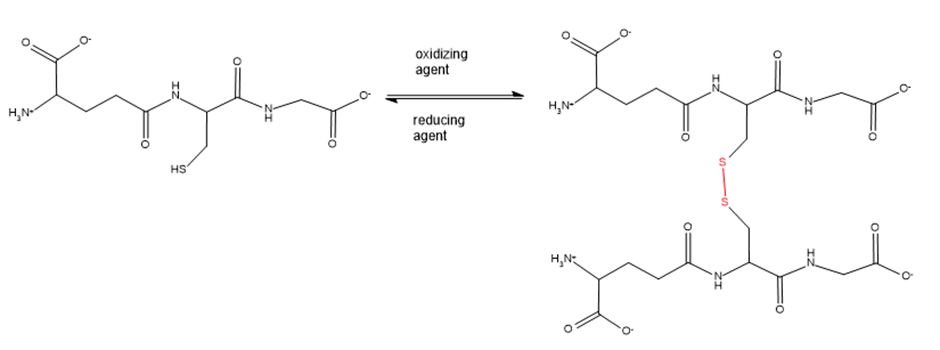
a. What amino acids make up glutathione?
b. What is unusual about glutathione’s structure?
alpha- Amino acids can be prepared by treating an aldehyde with ammonia/trace acid, followed by hydrogen cyanide, followed by acid-catalyzed hydrolysis.
a. Draw the structures of the two intermediates formed in this reaction.
b. What amino acid is formed when the aldehyde that is used is 3-methylbutanal?
c. What aldehyde is needed to prepare isoleucine?
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50 (Figure 21.3):
a. aspartate before serine c. valine before leucine
b. serine before alanine d. tyrosine before phenylalanine
What do you think about this solution?
We value your feedback to improve our textbook solutions.