Chapter 21: Q67P (page 1028)
Show how aspartame can be synthesized using DCCD.
Short Answer
The synthesis of aspartame using DCCD can be given as:
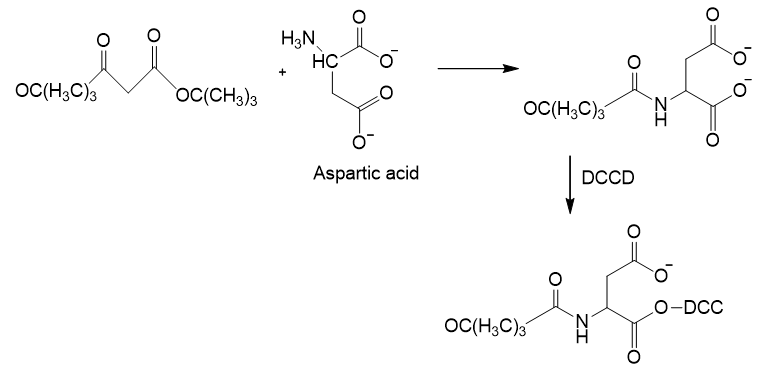
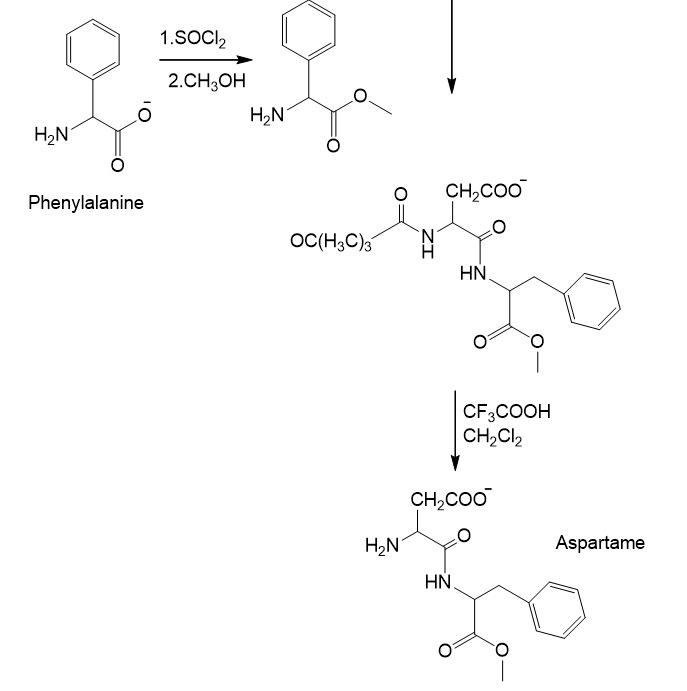
Learning Materials
Features
Discover
Chapter 21: Q67P (page 1028)
Show how aspartame can be synthesized using DCCD.
The synthesis of aspartame using DCCD can be given as:


All the tools & learning materials you need for study success - in one app.
Get started for free
Aspartame (its structure is on page 1007) has a pI of 5.9. Draw its prevailing form at physiological pH (7.4).
In what order would histidine, serine, aspartate, and valine be eluted with a buffer of pH 4 from a columncontaining an anion-exchange resin (Dowex 1)?
a. Which isomer—(R)-alanine or (S)-alanine—is d-alanine?
b. Which isomer—(R)-aspartate or (S)-aspartate—is d-aspartate?
c. Can a general statement be made relating R and S to d and l?
Why is excess ammonia used in the preceding reaction?
What amino acid is formed when the aldehyde used in the Strecker synthesis is
a. acetaldehyde? b. 2-methylbutanal? c. 3-methylbutanal?
What do you think about this solution?
We value your feedback to improve our textbook solutions.