Chapter 21: Q15P (page 997)
What aldehyde is formed when valine is treated with ninhydrin?
Short Answer
The aldehyde formed is 2-methyl propanal.
Learning Materials
Features
Discover
Chapter 21: Q15P (page 997)
What aldehyde is formed when valine is treated with ninhydrin?
The aldehyde formed is 2-methyl propanal.
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the mechanism for the reaction of a cysteine side chain with iodoacetic acid.
Which would have the greatest percentage of polar amino acids, a spherical protein, a cigar-shaped protein, or a subunit of a hexamer? b. Which would have the smallest percentage of polar amino acids?
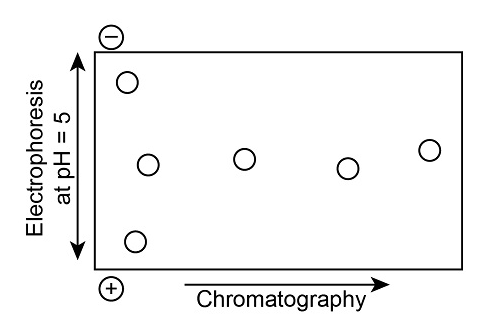
A mixture of amino acids that do not separate sufficiently when a single technique is used can often be separated by two-dimensional chromatography.In this technique, the mixture of amino acids is applied to a piece of filter paper and separated by chromatographic techniques. The paper is then rotated90°,and the amino acids are further separated by electrophoresis, producing a type of chromatogram called a fingerprint.Identify the spots in the fingerprint obtainedfrom a mixture of Ser, Glu, Leu, His, Met, and Thr.
Draw the predominant form for glutamate in a solution with the following pH:
a.0 b. 3 c. 6 d. 11
After the polypeptide shown below was treated with maleic anhydride, it was hydrolyzed by trypsin. (After a polypeptide is treated with maleic anhydride,
trypsin will cleave the polypeptide only on the C-side of arginine.)
Gly-Ala-Asp-Ala-Leu-Pro-Gly-Ile-Leu-Val-Arg-Asp-Val-Gly-Lys-Val-Glu-Val-Phe-Glu-Ala-Gly-
Arg-Ala-Glu-Phe-Lys-Glu-Pro-Arg-Leu-Val-Met-Lys-Val-Glu-Gly-Arg-Pro-Val-Gly-Ala-Gly-Leu-Trp
a. After a polypeptide is treated with maleic anhydride, why does trypsin no longer cleave it on the C-side of lysine?
b. How many fragments are obtained from the polypeptide?
c. In what order will the fragments be eluted from an anion-exchange column using a buffer of pH = 5?
What do you think about this solution?
We value your feedback to improve our textbook solutions.