Chapter 21: Q64P (page 1027)
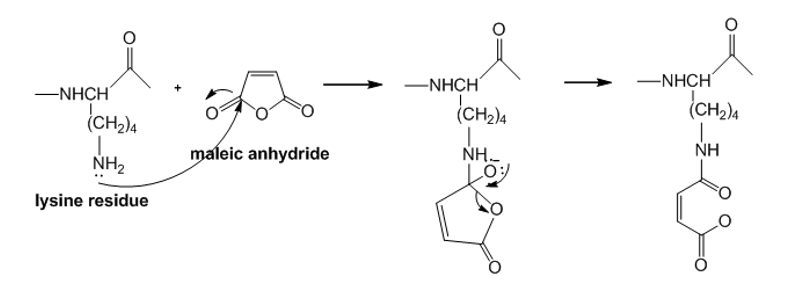
Draw the product obtained when a lysine side chain in a polypeptide reacts with maleic anhydride.

Short Answer
Learning Materials
Features
Discover
Chapter 21: Q64P (page 1027)
Draw the product obtained when a lysine side chain in a polypeptide reacts with maleic anhydride.


All the tools & learning materials you need for study success - in one app.
Get started for free
Reaction of a polypeptide with carboxypeptidase A releases Met. The polypeptide undergoes partial hydrolysis to give the following peptides.
What is the sequence of the polypeptide?
1. Ser, Lys, Trp4. Leu, Glu, Ser 7. Glu, His 10. Glu, His, Val
2. Gly, His, Ala 5. Met, Ala, Gly8. Leu, Lys, Trp11. Trp, Leu, Glu
3. Glu, Val, Ser 6. Ser, Lys, Val 9. Lys, Ser 12. Ala, Met
Draw the resonance contributors of the peptide bond in the less stable configuration.
a. What percentage of the -amino group of lysine will be protonated at its pI?<25%50% >75%
b. Answer the same question for the-amino group of lysine.
Draw the tetrapeptide Ala-Thr-Asp-Asn and indicate the peptide bonds.
Treatment of a polypeptide with 2-mercaptoethanol yields two polypeptides with the following primary structures:
Val-Met-Tyr-Ala-Cys-Ser-Phe-Ala-Glu-Ser
Ser-Cys-Phe-Lys-Cys-Trp-Lys-Tyr-Cys-Phe-Arg-Cys-Ser
Treatment of the original intact polypeptide with chymotrypsin yields the following peptides:
1. Ala, Glu, Ser 3. Tyr, Val, Met 5. Ser, Phe, 2 Cys, Lys, Ala, Trp
2. 2 Phe, 2 Cys, Ser 4. Arg, Ser, Cys 6. Tyr, Lys
Determine the positions of the disulfide bridges in the original polypeptide.
What do you think about this solution?
We value your feedback to improve our textbook solutions.