Chapter 21: Q13P (page 996)
a. What percentage of the -amino group of lysine will be protonated at its pI?<25%50% >75%
b. Answer the same question for the-amino group of lysine.
Short Answer
- >75%
- >75%
Learning Materials
Features
Discover
Chapter 21: Q13P (page 996)
a. What percentage of the -amino group of lysine will be protonated at its pI?<25%50% >75%
b. Answer the same question for the-amino group of lysine.
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the mechanism for the reaction of a cysteine side chain with iodoacetic acid.
Esterase is an enzyme that catalyzes the hydrolysis of esters. It hydrolyzes esters of L-amino acids more rapidly than esters of D-amino acids. How can this enzyme be used to separate a racemic mixture of amino acids?
Why are the carboxylic acid groups of the amino acids more acidic
(pKa ~ 2) than a carboxylic acid such as acetic acid (pKa = 4.76)?
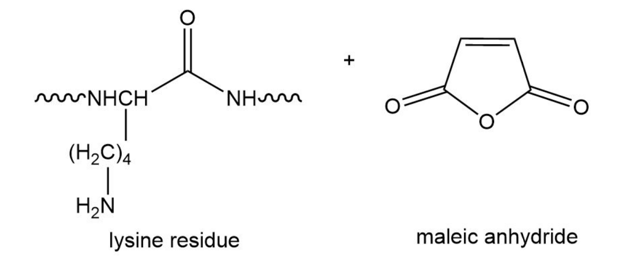
Draw the product obtained when a lysine side chain in a polypeptide reacts with maleic anhydride.
What amino acid is formed when the aldehyde used in the Strecker synthesis is
a. acetaldehyde? b. 2-methylbutanal? c. 3-methylbutanal?
What do you think about this solution?
We value your feedback to improve our textbook solutions.