Chapter 21: Q27P (page 1004)
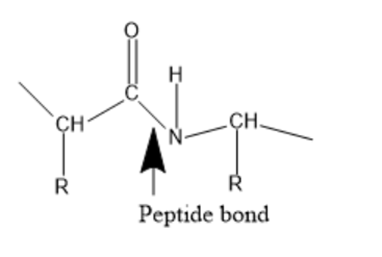
Draw the resonance contributors of the peptide bond in the less stable configuration.
Short Answer
Learning Materials
Features
Discover
Chapter 21: Q27P (page 1004)
Draw the resonance contributors of the peptide bond in the less stable configuration.

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50 (Figure 21.3):
a. aspartate before serine c. valine before leucine
b. serine before alanine d. tyrosine before phenylalanine
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)
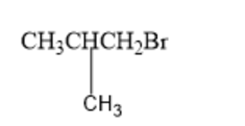
(b)
a.Which amino acid has the lowest pI value?
b.Which amino acid has the highest pI value?
c.Which amino acid has the greatest amount of negative charge at pH = 6.20?
d.Which amino acid has a greater negative charge at pH = 6.20, glycine or methionine?
Why are buffer solutions of increasingly higher pH used to elute the column that generates the chromatogram shown in Figure 21.5? (Elutemeans wash out with a solvent.)
Calculate the pI of each of the following amino acids:
a.asparagine b. arginine c. serine d. aspartate
What do you think about this solution?
We value your feedback to improve our textbook solutions.