Chapter 21: Q28P (page 1004)
Which bonds in the backbone of a peptide can rotate freely?
Short Answer

Learning Materials
Features
Discover
Chapter 21: Q28P (page 1004)
Which bonds in the backbone of a peptide can rotate freely?

All the tools & learning materials you need for study success - in one app.
Get started for free
A decapeptide undergoes partial hydrolysis to give peptides whose amino acid composition is shown. Reaction of the intact decapeptide with Edman’s reagent releases PTH-Gly. What is the sequence of the decapeptide?
1.Ala, Trp
2. Val,Pro,Asp
3. Pro, Val
4. Ala, Glu
5. Trp, Ala, Arg
6.Arg, Gly
7.Glu, Ala, Leu
8. Met, Pro, Leu, Glu
Draw the predominant form for glutamate in a solution with the following pH:
a.0 b. 3 c. 6 d. 11
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)
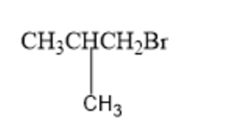
(b)
a. Calculate the overall yield of bradykinin when the yield for the addition of each amino acid to the chain is 70%.
b. What would be the overall yield of a peptide containing 15 amino acids if the yield for the incorporation of each is 80%?
Cells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism
(a) What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination?
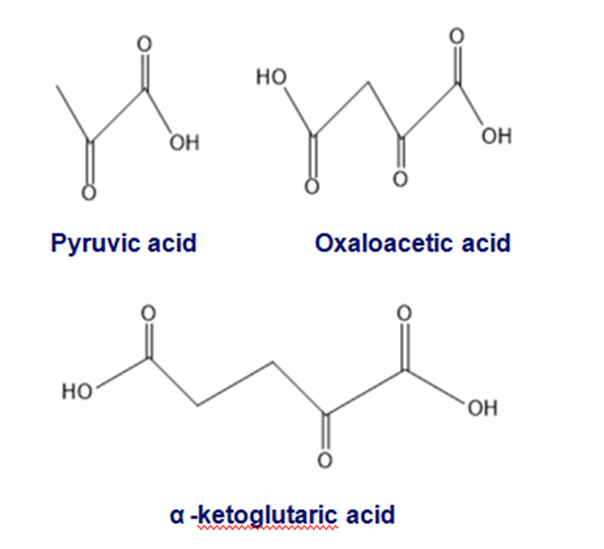
(b)What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?
What do you think about this solution?
We value your feedback to improve our textbook solutions.