Chapter 21: Q 22P (page 1002)
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)
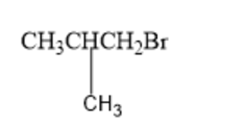
(b)
Short Answer
Answer:
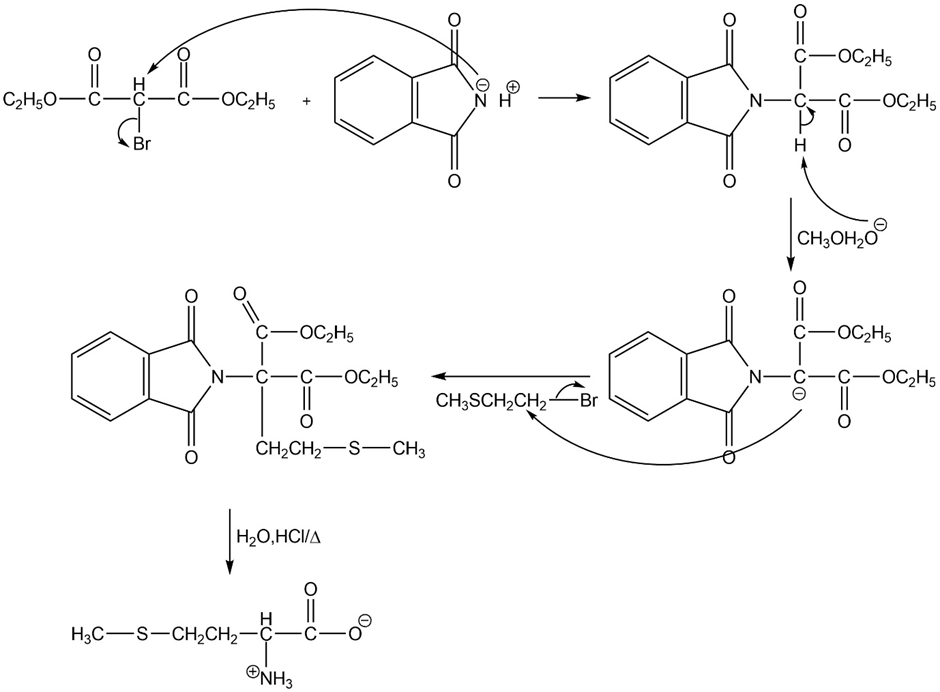
(a)

(b)
Learning Materials
Features
Discover
Chapter 21: Q 22P (page 1002)
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)

(b)
Answer:
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Which is the more effective buffer at physiological pH, a solution of 0.1 M glycylglycine or a solution of 0.2 M glycine?
In what order would histidine, serine, aspartate, and valine be eluted with a buffer of pH 4 from a columncontaining an anion-exchange resin (Dowex 1)?
Esterase is an enzyme that catalyzes the hydrolysis of esters. It hydrolyzes esters of L-amino acids more rapidly than esters of D-amino acids. How can this enzyme be used to separate a racemic mixture of amino acids?
Calculate the pI of each of the following amino acids:
a.asparagine b. arginine c. serine d. aspartate
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50 (Figure 21.3):
a. aspartate before serine c. valine before leucine
b. serine before alanine d. tyrosine before phenylalanine
What do you think about this solution?
We value your feedback to improve our textbook solutions.