Chapter 21: Q22P (page 1002)
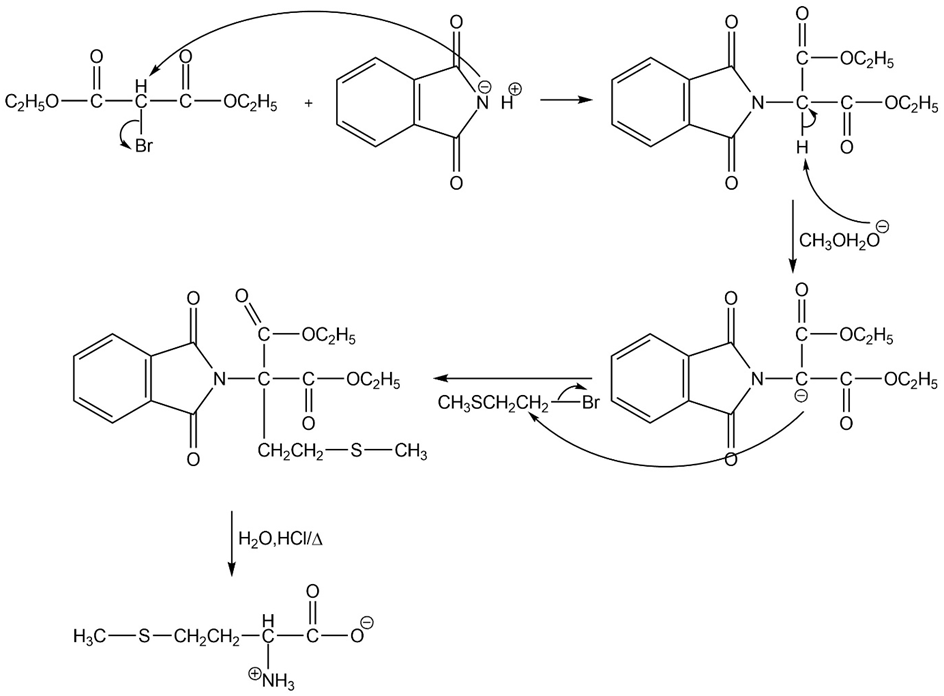
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)
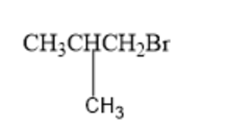
(b)
Short Answer
(a)
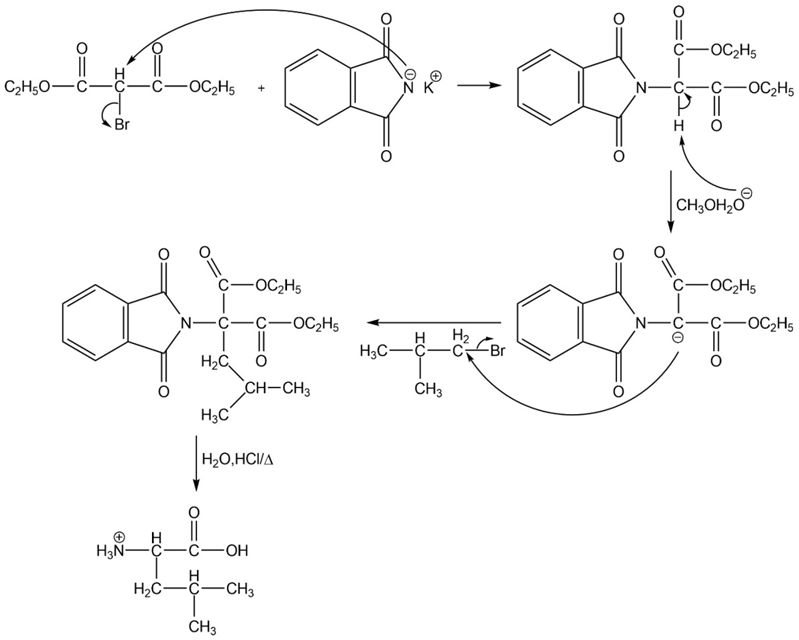
(b)
Learning Materials
Features
Discover
Chapter 21: Q22P (page 1002)
What amino acid is formed using the N-phthalimidomalonic ester synthesis when the following alkyl halides are used in the third step?
(a)

(b)
(a)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
Do any other amino acids in Table 21.2 have more than one asymmetric center?
Write the mechanism for the reaction of a cysteine side chain with iodoacetic acid.
Show how aspartame can be synthesized using DCCD.
Question: In determining the primary structure of insulin, what would lead you to conclude that insulin had more than one polypeptide chain?
What amino acid is formed when the aldehyde used in the Strecker synthesis is
a. acetaldehyde? b. 2-methylbutanal? c. 3-methylbutanal?
What do you think about this solution?
We value your feedback to improve our textbook solutions.