Chapter 21: Q10P (page 996)
Calculate the pI of each of the following amino acids:
a.asparagine b. arginine c. serine d. aspartate
Short Answer
(a) pI = 5.43
(b) pI = 10.76
(c) pI = 5.68
(d) pI = 2.98
Learning Materials
Features
Discover
Chapter 21: Q10P (page 996)
Calculate the pI of each of the following amino acids:
a.asparagine b. arginine c. serine d. aspartate
(a) pI = 5.43
(b) pI = 10.76
(c) pI = 5.68
(d) pI = 2.98
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the order of elution (with a buffer of pH 4) of the following pairs of amino acids through a column packed with Dowex 50 (Figure 21.3):
a. aspartate before serine c. valine before leucine
b. serine before alanine d. tyrosine before phenylalanine
What alkyl halide is used in the acetamidomalonic ester synthesis to prepare a. lysine? b. phenylalanine?
Draw the resonance contributors of the peptide bond in the less stable configuration.
a. What percentage of the -amino group of lysine will be protonated at its pI?<25%50% >75%
b. Answer the same question for the-amino group of lysine.
Glutathione is a tripeptide whose function is to destroy harmful oxidizing agents in the body. Oxidizing agents are bought to be responsible for some of the effects of aging and to play a causative role in cancer.
Glutathione removes oxidizing agents by reducing them. In the process, glutathione is oxidized, resulting in the formation of a disulphide bond between two glutathione molecules. An enzyme subsequently reduces the disulphide bond, returning glutathione to its original condition so it can react with another oxidizing agent.
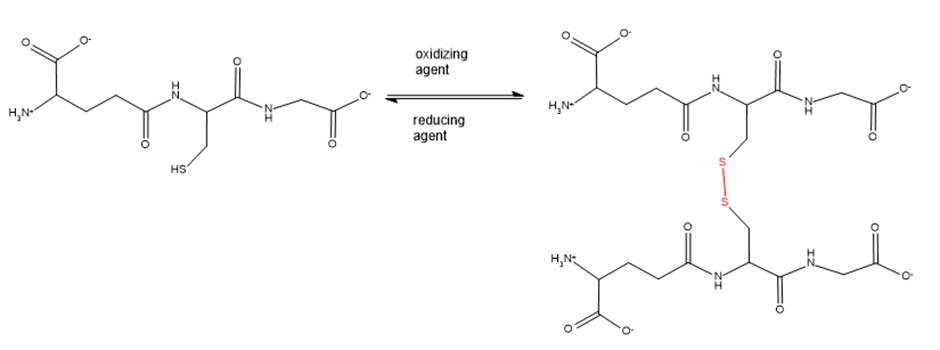
a. What amino acids make up glutathione?
b. What is unusual about glutathione’s structure?
What do you think about this solution?
We value your feedback to improve our textbook solutions.