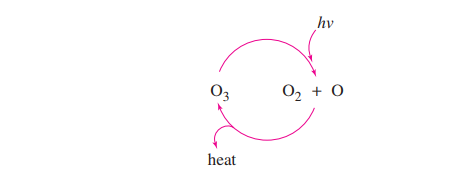
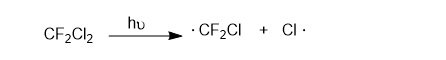When healthy, Earth’s stratosphere contains a low concentration of ozone (O3)that absorbs potentially harmful ultraviolet (UV) radiation by the cycle shown at right.
Chlorofluorocarbon refrigerants, such as Freon 12 (CF2Cl2), are stable in the lower atmosphere, but in the stratosphere, they absorb high-energy UV radiation to generate chlorine radicals.
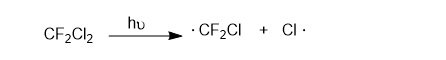
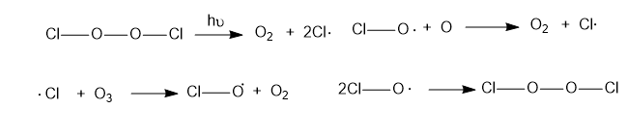
The presence of a small number of chlorine radicals appears to lower ozone concentrations dramatically. The following reactions are all known to be exothermic (except the one requiring light) and to have high-rate constants. Propose two mechanisms to explain how a small number of chlorine radicals can destroy large numbers of ozone molecules. Which of the two mechanisms is more likely when the concentration of chlorine atoms is very small?