Chapter 4: Q21P (page 215)
Predict the ratios of products that result from chlorination of isopentane.
Short Answer
The products formed will be in the ratio
Learning Materials
Features
Discover
Chapter 4: Q21P (page 215)
Predict the ratios of products that result from chlorination of isopentane.
The products formed will be in the ratio
All the tools & learning materials you need for study success - in one app.
Get started for free
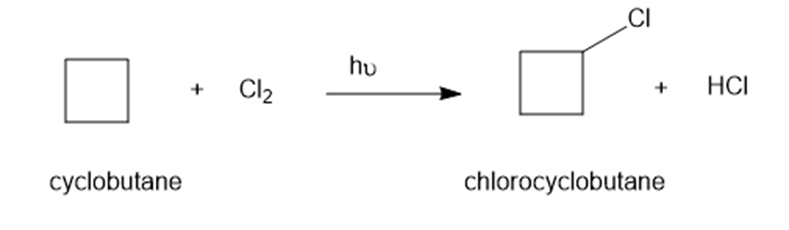
Write a mechanism for light-initiated reaction of cyclobutene with chlorine to give chlorocyclobutane. Label the initiation and propagation steps.
Peroxides are often added to free-radical reactions as initiators because the oxygen-oxygen bond cleaves homolytically rather easily. For example, the bond-dissociation enthalpy of O-Obond in hydrogen peroxide (H-O-O-H)is only 213 kJ/mol (51 kcal/mol). Give a mechanism for the hydrogen peroxide- initiated reaction of cyclopentane with chlorine. The BDE for HO-Clis 210 kJ/mol (50kcal/mol).
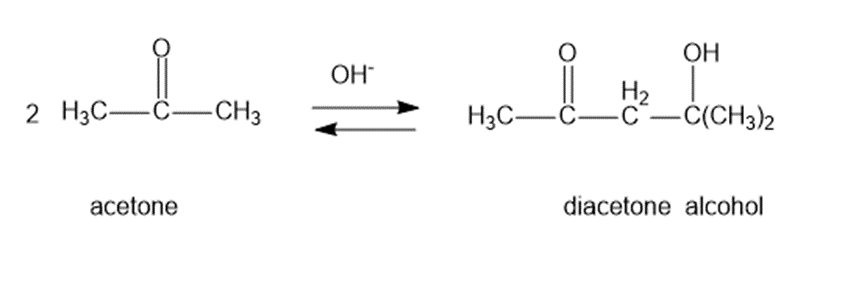
Question: Under base-catalyzed conditions, two molecules of acetone can condense to form diacetone alcohol. At room temperature, aboutof 5%the acetone is converted to diacetone alcohol. Determine the value offor this reaction.
Question: (a) Propose a mechanism for the free-radical chlorination of ethane.
(b) Calculatefor each step in this reaction
(c) Calculate the overall value offor this reaction.
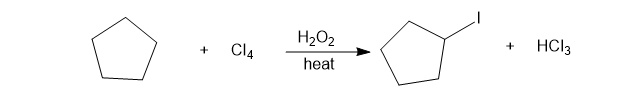
Iodination of alkanes using iodine (I2)is usually an unfavorable reaction. (See problem 4-17, for example). Tetraiodomethane (Cl4) can be used as the iodine source for iodination in the presence of a free-radical initiator such as hydrogen peroxide. Propose a mechanism (involving mildly exothermic propagation steps) for the following proposed reaction. Calculate the value of ��for each of the steps in your proposed mechanism.
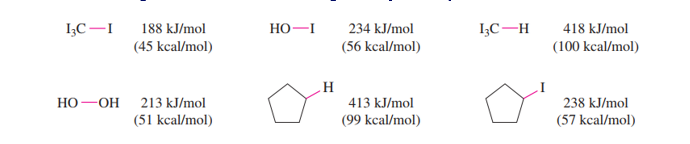
The following bond-dissociation energies may be helpful:
What do you think about this solution?
We value your feedback to improve our textbook solutions.