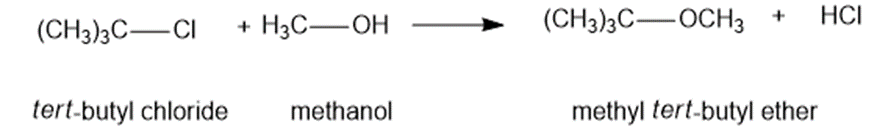
Chapter 4: Q6P (page 199)
Question: Under base-catalyzed conditions, two molecules of acetone can condense to form diacetone alcohol. At room temperature, aboutof 5%the acetone is converted to diacetone alcohol. Determine the value offor this reaction.

Short Answer
Answer
The value of is found to be 7.2KJ/mol.