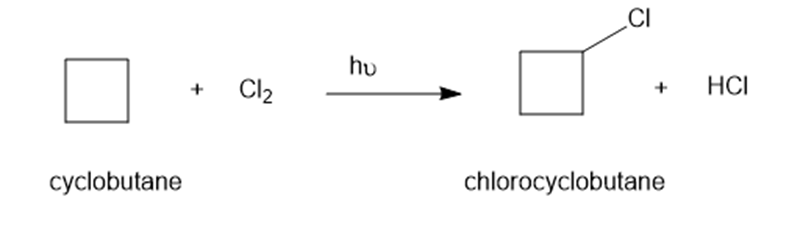
Chapter 4: Q7P (page 201)
Question: When ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs for this reaction. Explain these signs in terms of bonding and freedom of motion.
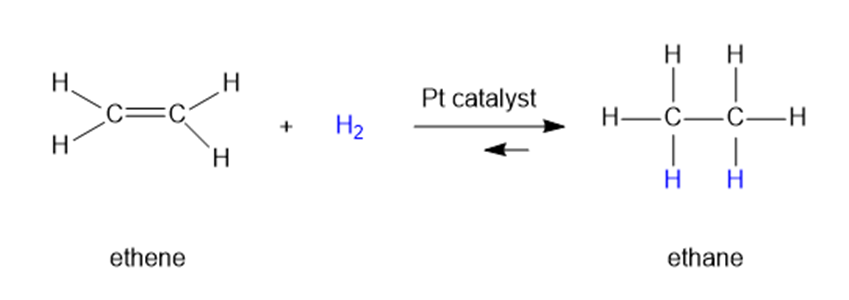
Short Answer
Answer
The value of is negative.
The value of is negative.