Chapter 4: Q 41P (page 234)
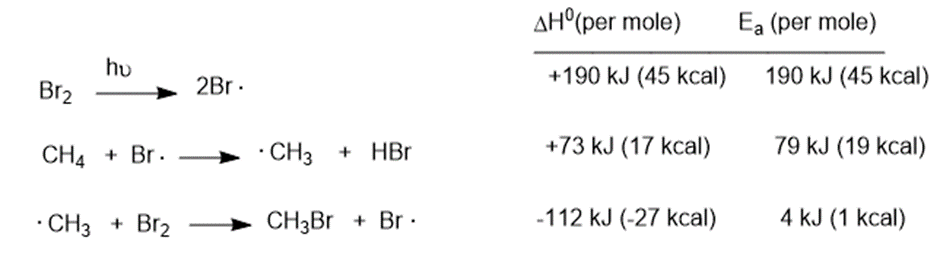
Use bond-dissociation enthalpies (Table 4-2, p.203) to calculate values ofΔHfor the following reactions.
(a)

(b)

(c)
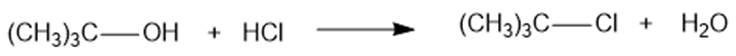
(d)

(e)

Short Answer
(a) ΔH = -36kJ/mol
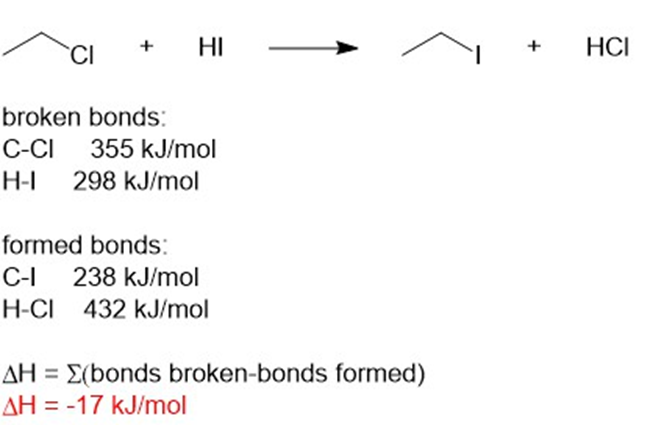
(b) ΔH = -17kJ/mol
(c) ΔH = -19kJ/mol
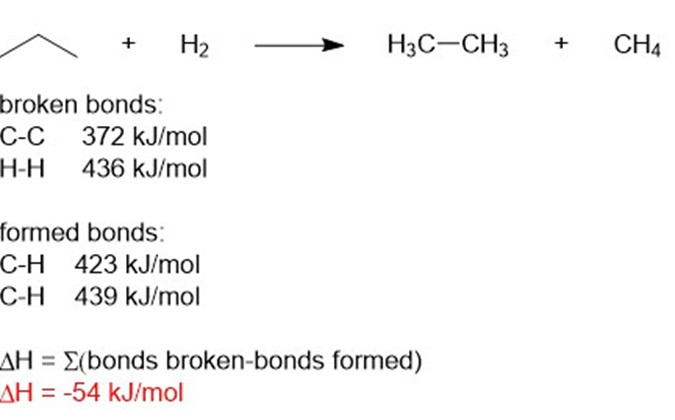
(d) ΔH= -54kJ/mol
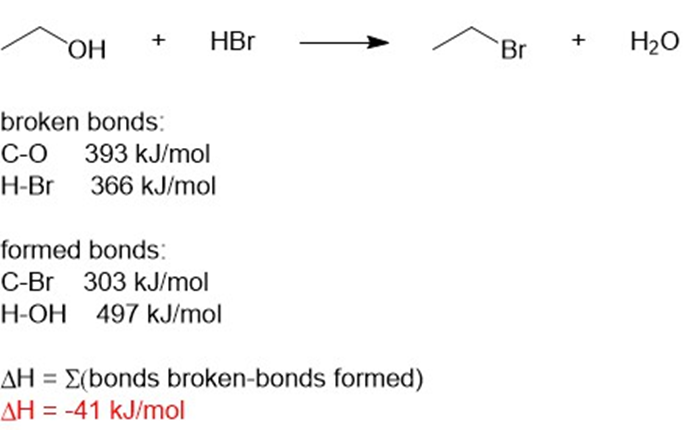
(e) ΔH = -41kJ/mol