Chapter 4: Q 44P (page 234)
Write a mechanism for light-initiated reaction of cyclobutene with chlorine to give chlorocyclobutane. Label the initiation and propagation steps.
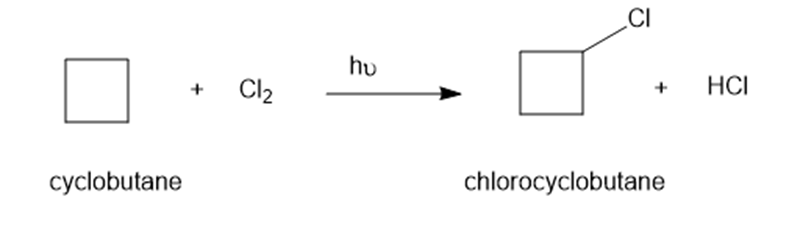
Short Answer
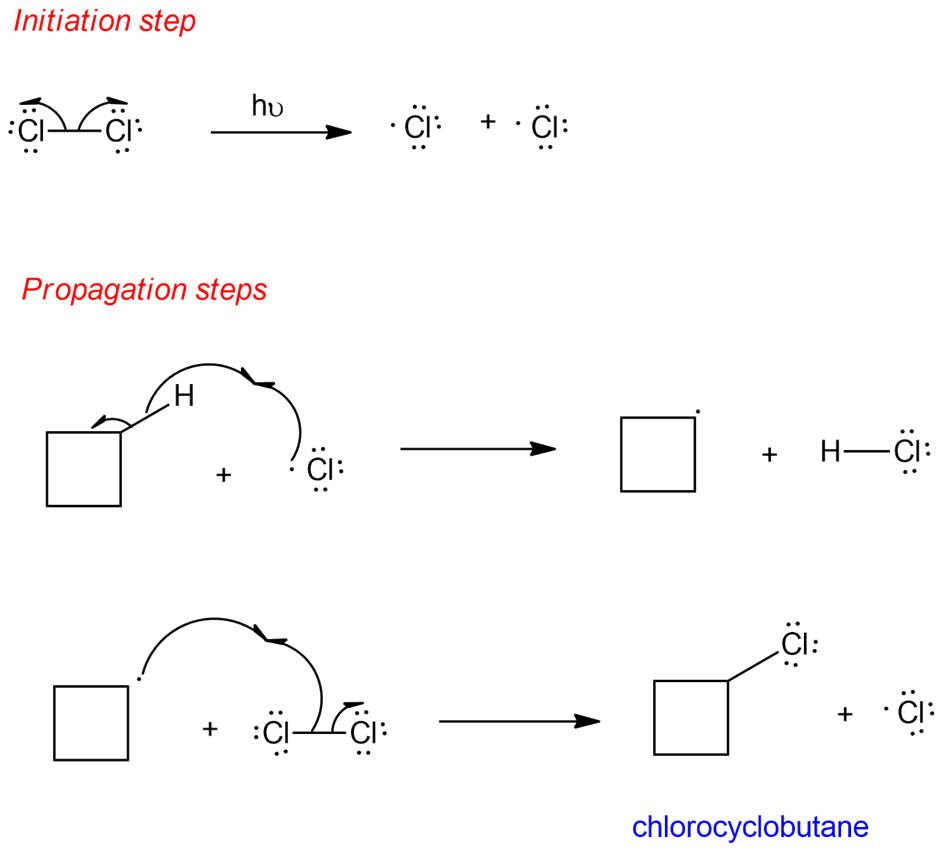
Mechanism for free-radical chlorination of cyclobutane
Learning Materials
Features
Discover
Chapter 4: Q 44P (page 234)
Write a mechanism for light-initiated reaction of cyclobutene with chlorine to give chlorocyclobutane. Label the initiation and propagation steps.


Mechanism for free-radical chlorination of cyclobutane
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: For each reaction, estimate whether for the reaction is positive, negative or impossible to predict.
a.
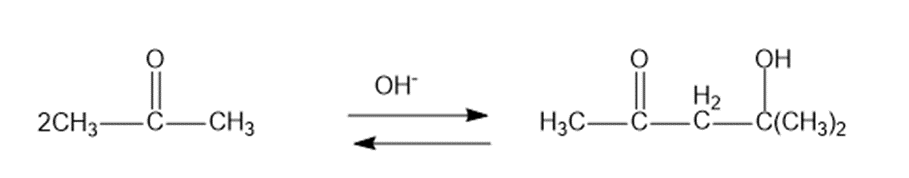
b.The formation of diacetone alcohol:
c.
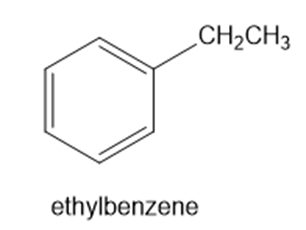
For each compound, predict the major product of free-radical bromination. Remember that bromination is highly selective, and only the most stable radical will be formed.
(a) cyclohexane
(b) methylcyclopentane
(c) decalin
(d) hexane
(e)
(f)
Question: Free-radical chlorination of hexane gives very poor yields of 1 clorohexyane, while cyclohexane can be converted to chlorocyclohexane in good yield.
(a)How do you account for this difference?
(b) What ratio of reactants (cyclohexane and chlorine)
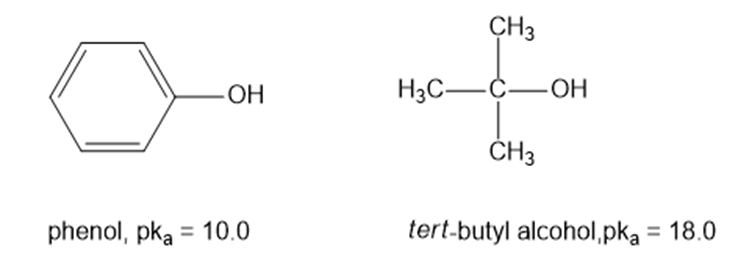
(a) Draw an approximate reaction-energy diagram for the acid-base reaction of phenol (see below) with 1-molar aqueous sodium hydroxide solution.
(b) On the same diagram, draw an approximate reaction-energy diagram for the acid-base reaction oftert- butyl alcohol(see below) with 1-molar aqueous sodium hydroxide solution.
Question: (a) Use bond-dissociation enthalpies from Table 4-2 (page 203), calculate the heat of reaction for each step in the free-radical bromination of methane.

(b) Calculate the overall heat of reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.