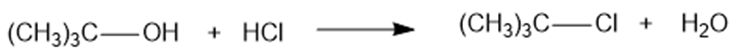Chapter 4: Q 38P (page 233)
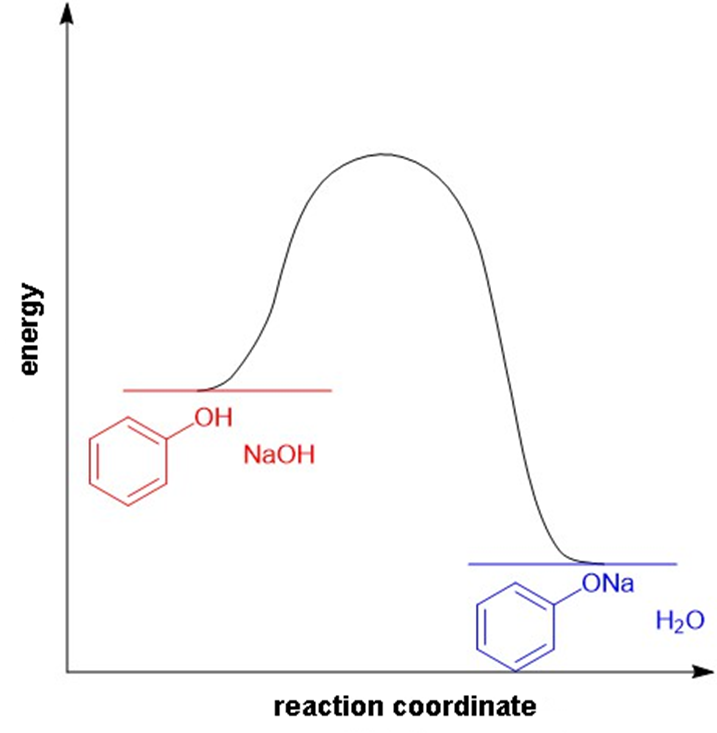
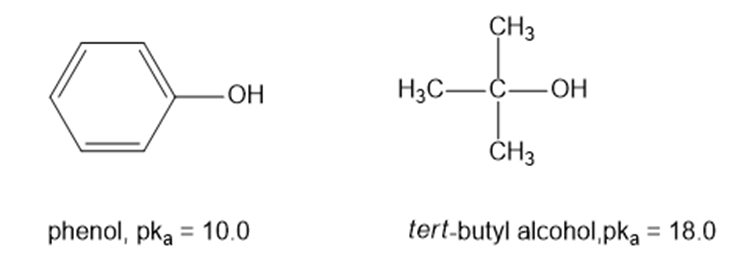
(a) Draw an approximate reaction-energy diagram for the acid-base reaction of phenol (see below) with 1-molar aqueous sodium hydroxide solution.
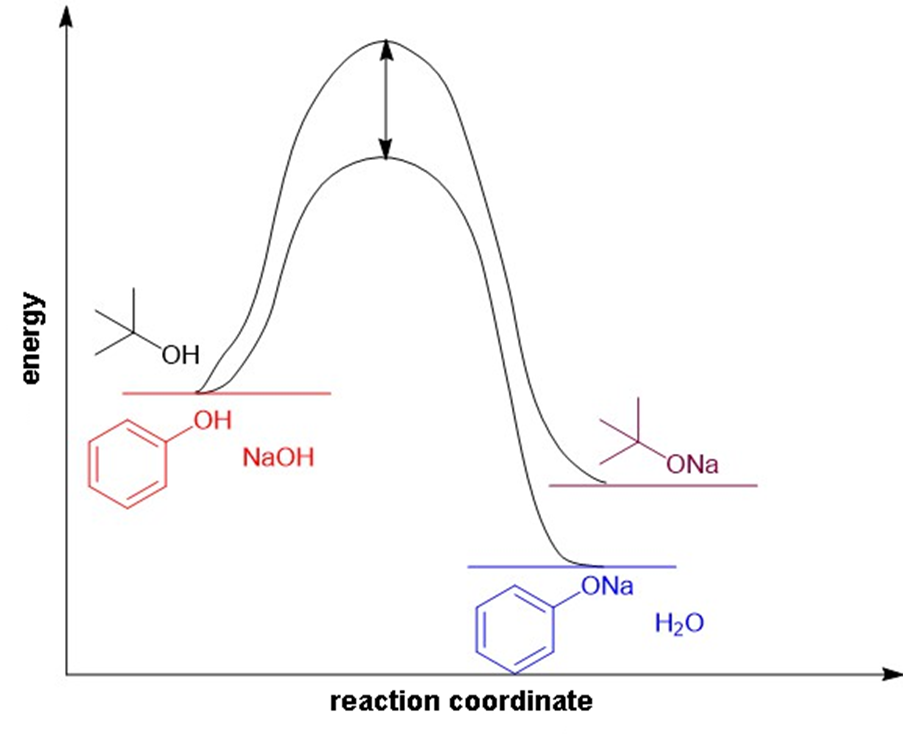
(b) On the same diagram, draw an approximate reaction-energy diagram for the acid-base reaction oftert- butyl alcohol(see below) with 1-molar aqueous sodium hydroxide solution.
Short Answer
(a)
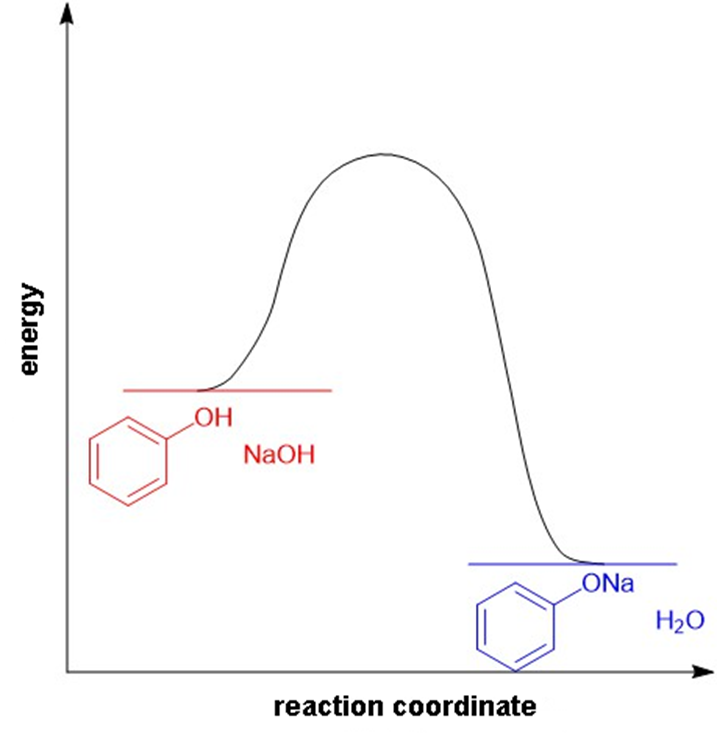
Reaction-energy diagram for the acid-base reaction of phenol with 1-molar aqueous sodium hydroxide solution.
(b)
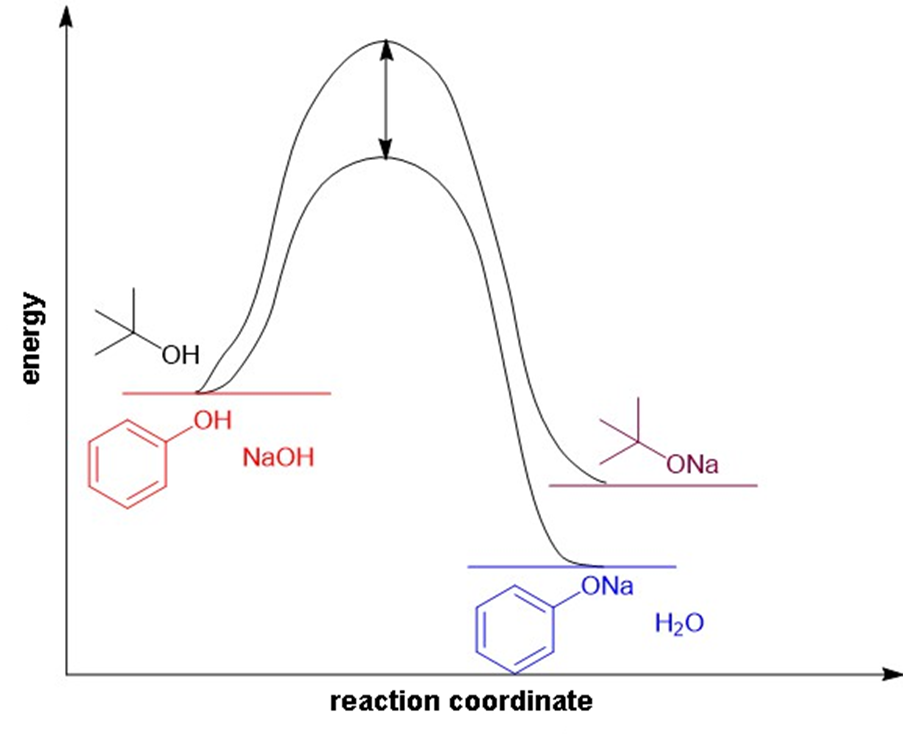
Reaction-energy diagram for the acid-base reaction of and phenol with 1-molar aqueous sodium hydroxide solution.