Chapter 4: Q 37P (page 233)
Draw a reaction-energy diagram for a two-step endothermic reaction with a rate-limiting step.
Short Answer
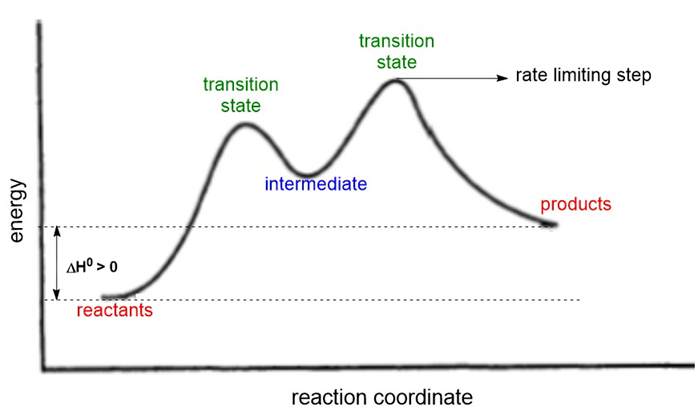
reaction-energy diagram for a two-step endothermic reaction with a rate-limiting step
Learning Materials
Features
Discover
Chapter 4: Q 37P (page 233)
Draw a reaction-energy diagram for a two-step endothermic reaction with a rate-limiting step.

reaction-energy diagram for a two-step endothermic reaction with a rate-limiting step
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The bromination of methane proceeds through the following steps:
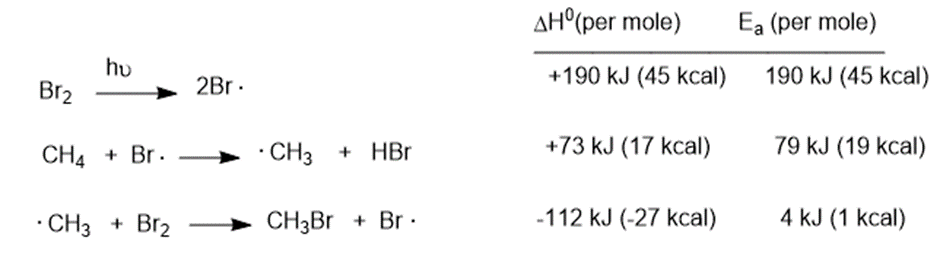
(a) Draw a complete reaction-energy diagram for this reaction.
(b) Label the rate-limiting step.
(c) Draw the structure of each transition state
(d) Compute the overall value offor the bromination.
What would be the product ratio in the chlorination of propane if all the hydrogens were abstracted at equal rates?
Label each hydrogen atom in the following compound as primary (1o), secondary (2o), or tertiary (3o)
(a)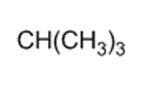
(b)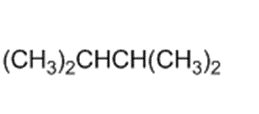
(c)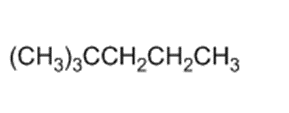
(d)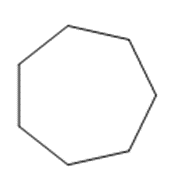
e)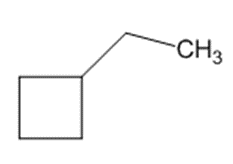
(f)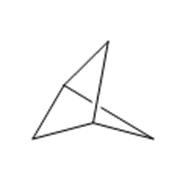
In the presence of a small amount of bromine, cyclohexene undergoes the following light-promoted reaction:
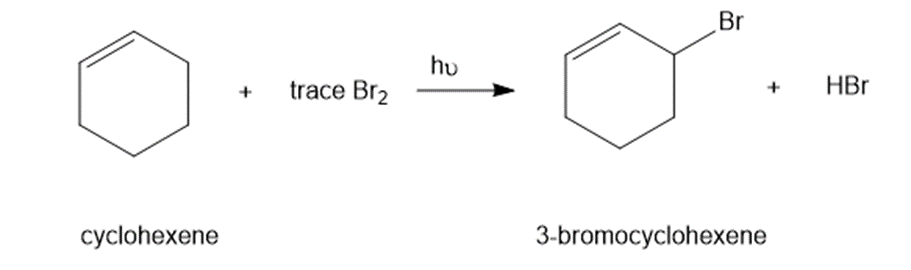
(a) Propose a mechanism for this reaction.
(b) Draw the structure of the rate-limiting transition state.
(c) Use Hammond’s postulate to predict which intermediate most closely resembles this transition state.
(d) Explain why cyclohexene reacts with bromine much faster than cyclohexane, which must be heated to react.
Question: (a) Use bond-dissociation enthalpies from Table 4-2 (page 203), calculate the heat of reaction for each step in the free-radical bromination of methane.

(b) Calculate the overall heat of reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.