Chapter 4: Q-4-53P (page 235)
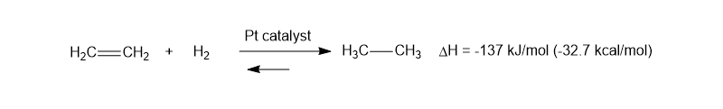
When ethene is treated in a calorimeter with H2and a Ptcatalyst, the heat of reaction is found to be -137kJ/mol(-32.7kcal/mol), and the reaction goes to completion. When the reaction takes place at 14000K, the equilibrium is found to be evenly balanced, with Keq=1. Compute the value of ��for this reaction.
Short Answer
�� = -98J/mol.K


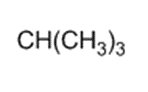
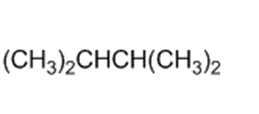
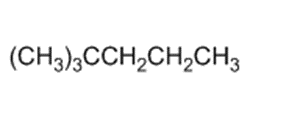
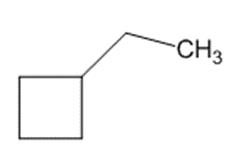
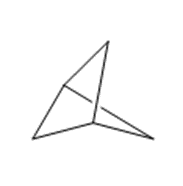


 )is deprotonated by very strong bases. Write resonance forms to show the stabilization of the carbanion that results.
)is deprotonated by very strong bases. Write resonance forms to show the stabilization of the carbanion that results.