Chapter 4: Q27P (page 223)
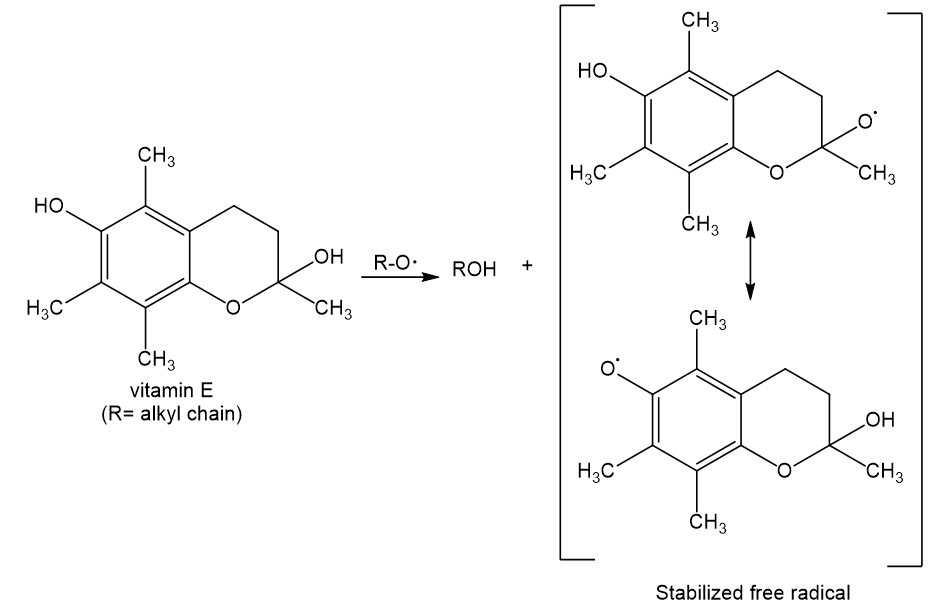
Write an equation for the reaction of vitamin E with an oxidizing radical (RO·) to give ROH and a less reactive free radical.
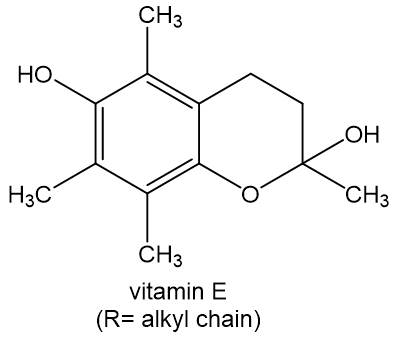
Short Answer
Learning Materials
Features
Discover
Chapter 4: Q27P (page 223)
Write an equation for the reaction of vitamin E with an oxidizing radical (RO·) to give ROH and a less reactive free radical.


All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Draw the structure of the transition state for the second propagation step in the chlorination of methane.
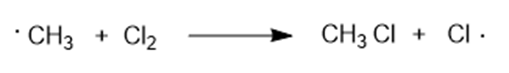
Show whether the transition state is product-like or reactant-like and which of the two partial bonds is stronger.
(b) Repeat for the second propagation step in the bromination of methane.
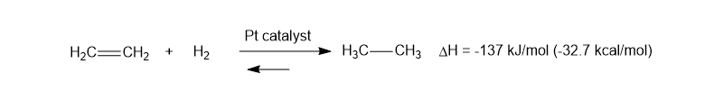
When ethene is treated in a calorimeter with H2and a Ptcatalyst, the heat of reaction is found to be -137kJ/mol(-32.7kcal/mol), and the reaction goes to completion. When the reaction takes place at 14000K, the equilibrium is found to be evenly balanced, with Keq=1. Compute the value of ��for this reaction.
(a) Compute the heats of reaction for abstraction of a primary hydrogen and a secondary hydrogen from propane by a fluorine radical.

(b) How selective do you expect free-radical fluorination to be?
(c) What product distribution you expect to obtain from the free-radical fluorination of propane?
Question: Draw Lewis structures for the following free radicals.
(a)The ethyl radical, CH3-CH2
(b)The tert butyl radical, (CH3 )3C
(c)The isopropyl radical,(2-propyl radical)
(d)The iodine atom
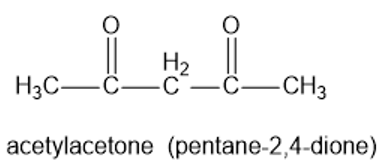
Acetylacetone (pentane -2, 4-dione)reacts with sodium hydroxide to give water and the sodium salt of a carbanion. Write a complete structural formula for the carbanion, and use resonance forms to show the stabilization of the carbanion.
What do you think about this solution?
We value your feedback to improve our textbook solutions.