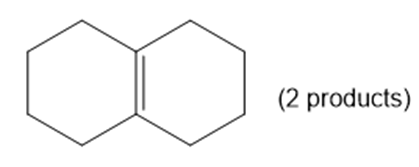
Chapter 4: Q23-P (page 219)
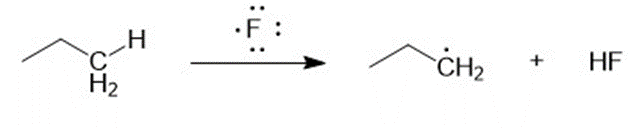
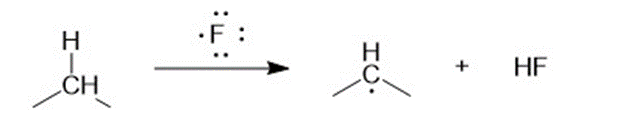
(a) Compute the heats of reaction for abstraction of a primary hydrogen and a secondary hydrogen from propane by a fluorine radical.
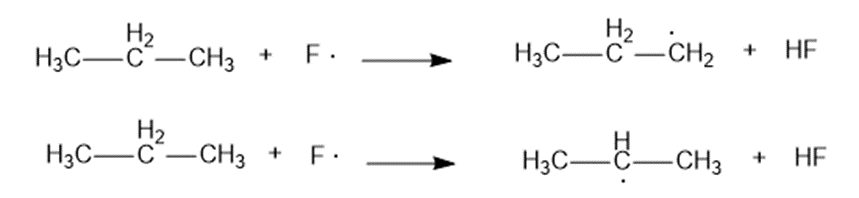
(b) How selective do you expect free-radical fluorination to be?
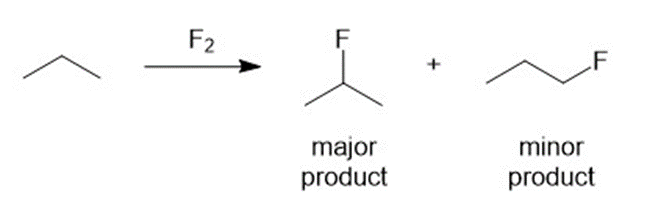
(c) What product distribution you expect to obtain from the free-radical fluorination of propane?
Short Answer
(a) For abstraction of primary hydrogen by a fluorine radical,
For abstraction of secondary hydrogen by a fluorine radical,
(b) Free-radical fluorination is nearly non-selective.
(c) The second reaction has more negative change in enthalpy which implies that it is more feasible. Secondary carbon is more easily fluorinated.