Chapter 4: Q2P (page 195)
Question: (a) Write the propagation steps leading to the formation of dichloromethane (CH2Cl2).
(b) Explain why free-radical halogenation usually gives mixture of products.
(c) How could an industrial plant control the proportions of methane and chlorine to favor production of CCl4? To favor production of CH3Cl?
Short Answer
Answer
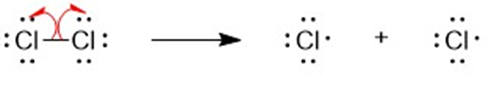
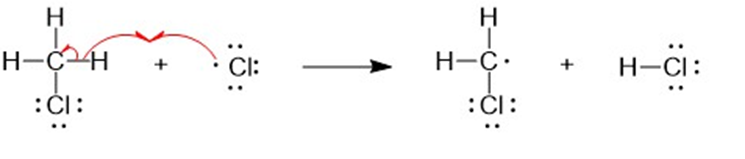
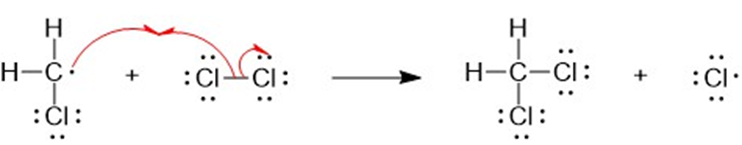
a
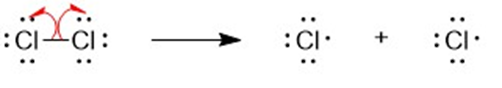
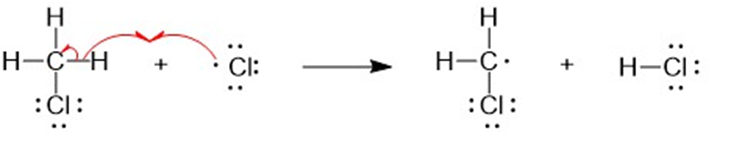
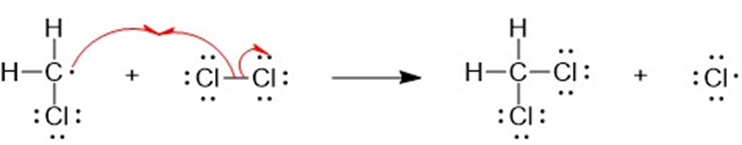
(b) A halogen atom is substituted for a hydrogen atom in free-radical halogenation. A different compound is produced from the first substitution of a hydrogen atom. The hydrogen atoms left unreacted can compete for halogen atoms that are available. For example, in the chlorination of methane, different substitution products are formed such as CH4,CH3Cl,CH2CI2, CHCI3, and CCI4. Hence, free-radical halogenation usually gives a mixture of products.
(c)The amount ofCI2 (chlorine) reacted with CH4 (methane) should be in excess for favoring the production ofCCI4 . This is to make certain that the reaction progresses so that all the C-H bonds are replaced with C-l bonds.
The amount ofCI2 (chlorine) reacted withCH4 (methane) should be less to favor the production of . This is to make certain that the reaction progresses so that all the chlorine atoms would more likely react with methane to formCH3Cl and not with CH3Cl to form CH2CI2 .