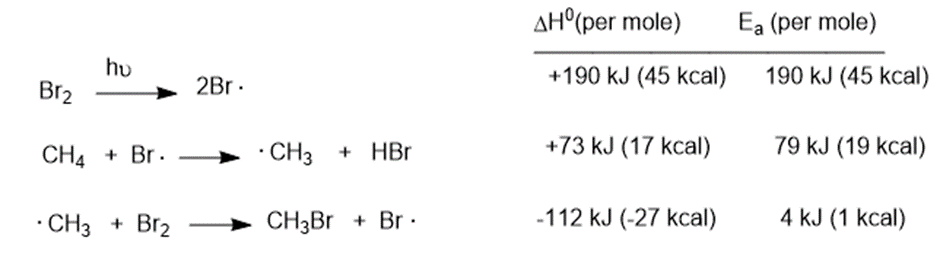
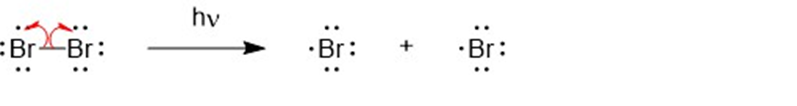Chapter 4: Q24P (page 221)
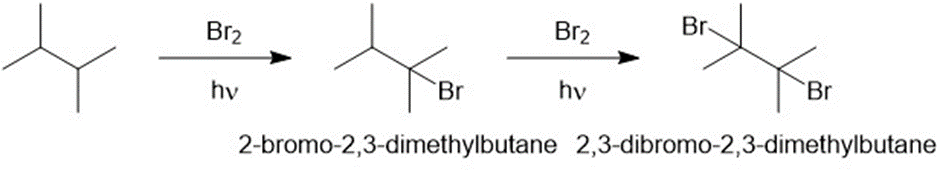
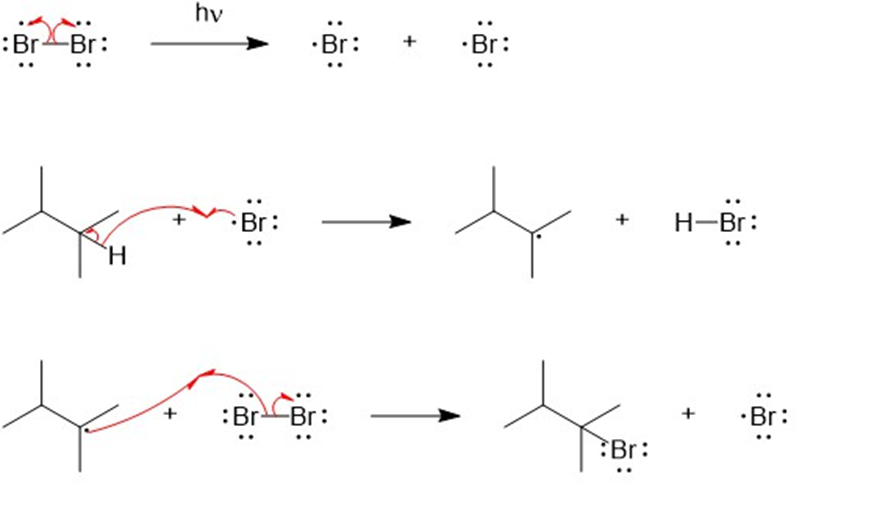
reacts with bromine in the presence of light to give a mono brominate product. Further reaction gives a good yield of a dibrominated product. Predict the structures of these products, and propose a mechanism for the formation of the monobrominated product.
Short Answer
The Themonobrominated product formed is 2-bromo-2, 3-dimethylbutane and the dibrominated product formed is 2, 3-dibromo-2, 3-dimethylbutane.
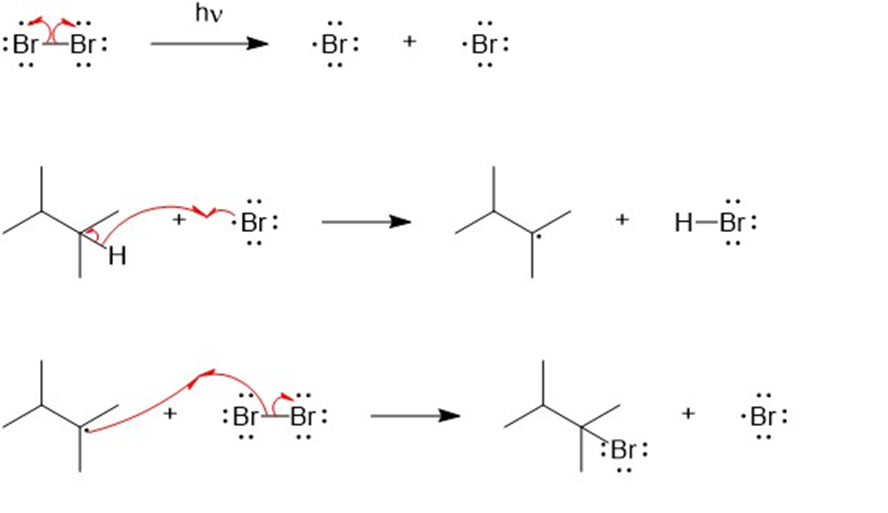
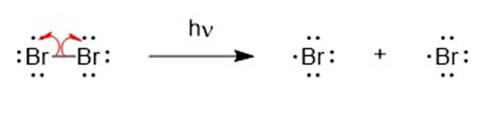
The mechanism for the formation of mono brominated product.
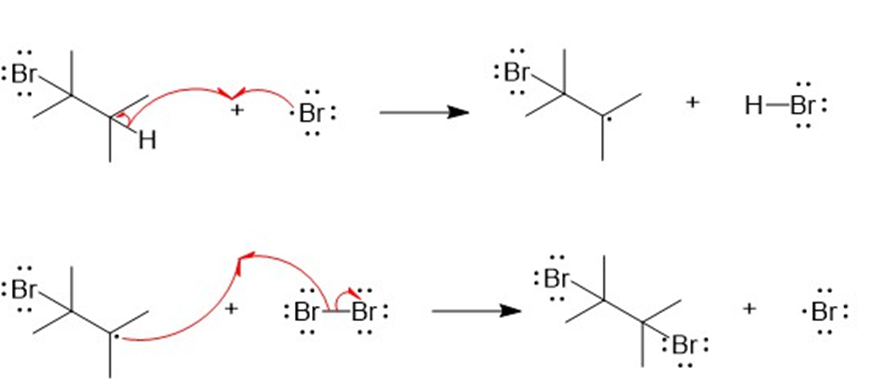
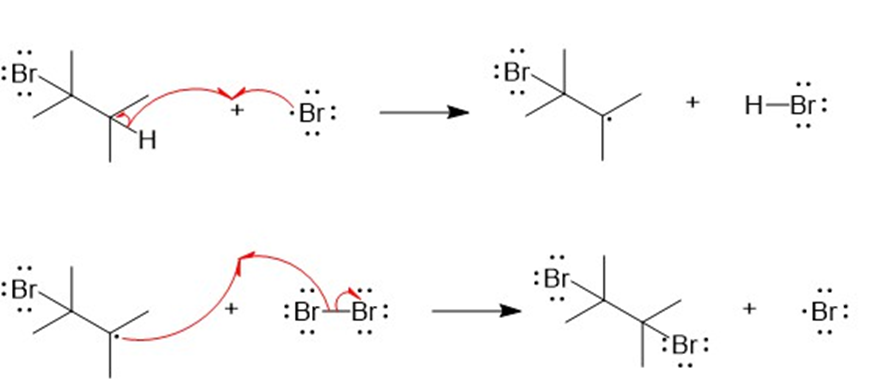
The mechanism for the formation of dibrominated product