Chapter 4: Q33P (page 233)
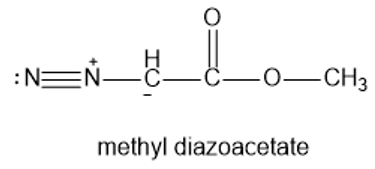
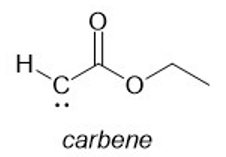
When it is strongly heated, methyl diazoacetate decomposes to give a nitrogen gas and a carbene. Draw a Lewis structure of the carbene.
Short Answer
Learning Materials
Features
Discover
Chapter 4: Q33P (page 233)
When it is strongly heated, methyl diazoacetate decomposes to give a nitrogen gas and a carbene. Draw a Lewis structure of the carbene.


All the tools & learning materials you need for study success - in one app.
Get started for free
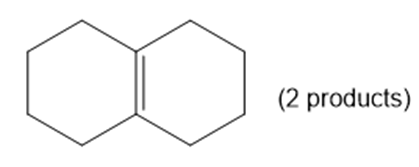
For each compound, predict the major product of free-radical bromination. Remember that bromination is highly selective, and only the most stable radical will be formed.
(a) cyclohexane
(b) methylcyclopentane
(c) decalin
(d) hexane
(e)
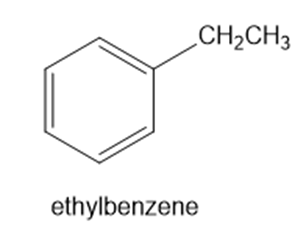
(f)
Propionitrile( )is deprotonated by very strong bases. Write resonance forms to show the stabilization of the carbanion that results.
)is deprotonated by very strong bases. Write resonance forms to show the stabilization of the carbanion that results.
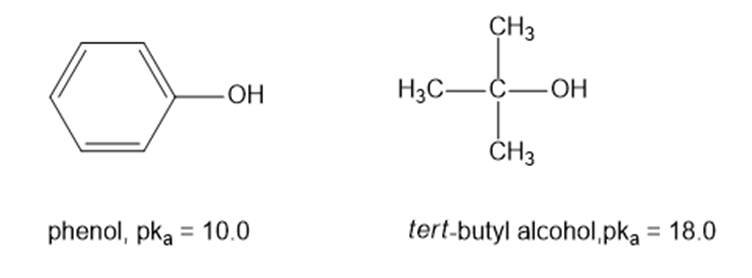
(a) Draw an approximate reaction-energy diagram for the acid-base reaction of phenol (see below) with 1-molar aqueous sodium hydroxide solution.
(b) On the same diagram, draw an approximate reaction-energy diagram for the acid-base reaction oftert- butyl alcohol(see below) with 1-molar aqueous sodium hydroxide solution.
In the presence of a small amount of bromine, cyclohexene undergoes the following light-promoted reaction:
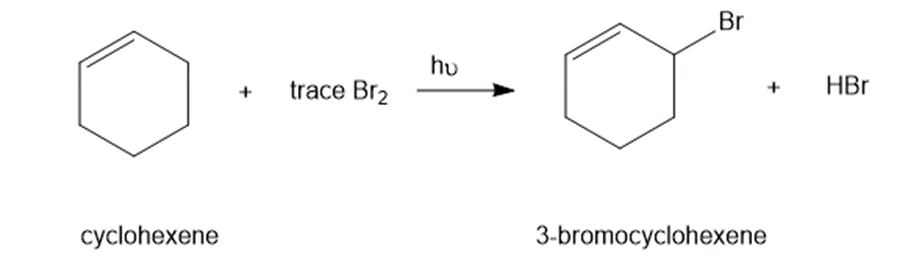
(a) Propose a mechanism for this reaction.
(b) Draw the structure of the rate-limiting transition state.
(c) Use Hammond’s postulate to predict which intermediate most closely resembles this transition state.
(d) Explain why cyclohexene reacts with bromine much faster than cyclohexane, which must be heated to react.
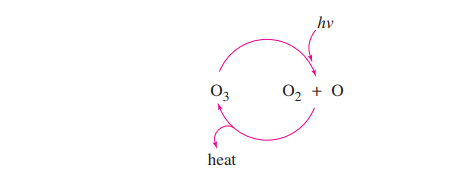
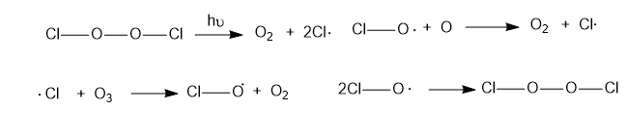
When healthy, Earth’s stratosphere contains a low concentration of ozone (O3)that absorbs potentially harmful ultraviolet (UV) radiation by the cycle shown at right.
Chlorofluorocarbon refrigerants, such as Freon 12 (CF2Cl2), are stable in the lower atmosphere, but in the stratosphere, they absorb high-energy UV radiation to generate chlorine radicals.
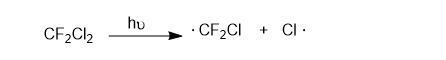
The presence of a small number of chlorine radicals appears to lower ozone concentrations dramatically. The following reactions are all known to be exothermic (except the one requiring light) and to have high-rate constants. Propose two mechanisms to explain how a small number of chlorine radicals can destroy large numbers of ozone molecules. Which of the two mechanisms is more likely when the concentration of chlorine atoms is very small?
What do you think about this solution?
We value your feedback to improve our textbook solutions.